Electron Configuration Practice Questions with Answers
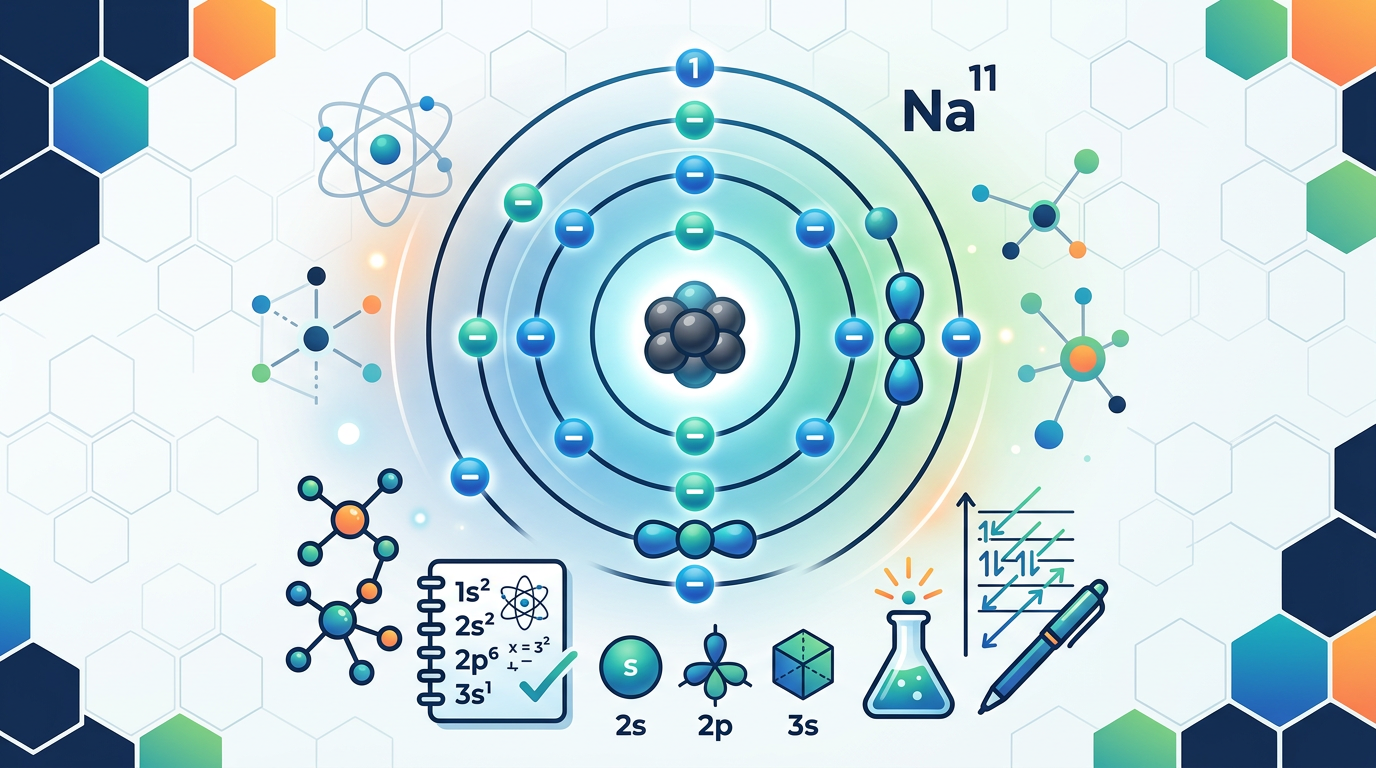
Concept Explanation
Electron configuration is the symbolic representation of how an atom's electrons are distributed among the various atomic orbitals and shells according to their energy levels. This distribution follows the laws of quantum mechanics to ensure the atom is in its lowest energy state, also known as the ground state. Understanding this concept is fundamental for predicting chemical reactivity, bonding behavior, and the magnetic properties of elements. To determine the electron configuration of an atom, scientists follow three primary principles: the Aufbau Principle, which states electrons fill lower energy orbitals first; Hund’s Rule, which suggests electrons fill degenerate orbitals singly before pairing; and the Pauli Exclusion Principle, which dictates that no two electrons in an atom can have the same four quantum numbers.
The notation typically consists of the principal energy level (n), the orbital type (s, p, d, or f), and a superscript indicating the number of electrons in that subshell. For example, the notation 1s² indicates two electrons in the s-orbital of the first energy level. As elements become more complex, chemists often use the "Noble Gas Notation" to shorten the string by using the preceding noble gas to represent the core electrons. Much like how redox reaction practice questions help you master electron transfer between atoms, electron configuration helps you understand where those electrons reside before any reaction occurs. For more in-depth study of atomic behavior, resources like the Khan Academy Electronic Structure or the IUPAC nomenclature guides provide extensive technical background.
Key Rules for Electron Filling
-
Aufbau Principle: Electrons occupy the lowest energy orbital available. The sequence generally follows the 1s, 2s, 2p, 3s, 3p, 4s, 3d order.
-
Pauli Exclusion Principle: An orbital can hold a maximum of two electrons, and they must have opposite spins.
-
Hund's Rule: For orbitals of the same energy (like the three 2p orbitals), electrons will fill them one by one with parallel spins before they start to pair up.
Solved Examples
Below are three fully worked examples showing the step-by-step process of determining electron configurations for different elements.
-
Determine the full electron configuration for Phosphorus (Z = 15).
-
Identify the total number of electrons: Phosphorus has 15 electrons.
-
Start filling from the lowest energy level: 1s can hold 2 electrons → 1s².
-
Move to the next level: 2s can hold 2 electrons → 2s².
-
Move to the 2p subshell: 2p can hold 6 electrons → 2p⁶ (Total used: 10).
-
Move to the 3s subshell: 3s can hold 2 electrons → 3s² (Total used: 12).
-
The remaining 3 electrons go into the 3p subshell → 3p³.
-
Final Configuration: 1s² 2s² 2p⁶ 3s² 3p³.
-
-
Write the noble gas notation for Iron (Z = 26).
-
Identify the noble gas that precedes Iron: Argon (Z = 18).
-
Write the symbol for Argon in brackets: [Ar]. This represents the first 18 electrons (1s² 2s² 2p⁶ 3s² 3p⁶).
-
Determine the remaining electrons: 26 - 18 = 8 electrons.
-
Fill the 4s subshell first (as per Aufbau): 4s².
-
The remaining 6 electrons fill the 3d subshell: 3d⁶.
-
Final Notation: [Ar] 4s² 3d⁶.
-
-
Determine the electron configuration for the Copper (Cu) atom (Z = 29), noting any exceptions.
-
Identify total electrons: 29.
-
Expected configuration based on rules: [Ar] 4s² 3d⁹.
-
Apply the stability exception: Half-filled and fully-filled d-subshells are more stable. An electron moves from 4s to 3d.
-
Adjusted configuration: [Ar] 4s¹ 3d¹⁰.
-
Final Configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹ 3d¹⁰.
-
Practice Questions
Test your knowledge with these electron configuration practice questions ranging from basic to advanced levels.
1. Write the complete electron configuration for the Fluorine atom (Z = 9).
2. Identify the element that has the ground-state electron configuration: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s².
3. Write the noble gas notation for the element Bromine (Z = 35).
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. Determine the electron configuration for the Sodium ion (Na⁺). Note that Sodium has an atomic number of 11.
5. Which element has the valence shell configuration of 5s² 5p⁴?
6. Write the full electron configuration for Chromium (Z = 24), being mindful of the stability rules for d-orbitals.
7. How many unpaired electrons are present in a neutral Nitrogen atom (Z = 7)?
8. Write the electron configuration for the Sulfide ion (S²⁻). Sulfur has 16 protons.
9. Identify the element with the configuration [Kr] 5s² 4d¹⁰ 5p².
10. Explain why the 4s orbital fills before the 3d orbital in the third and fourth periods.
Answers & Explanations
-
1s² 2s² 2p⁵: Fluorine has 9 electrons. Two go to 1s, two to 2s, and the remaining five fill the 2p subshell.
-
Calcium (Ca): Adding the superscripts (2+2+6+2+6+2) gives 20. Element 20 on the periodic table is Calcium.
-
[Ar] 4s² 3d¹⁰ 4p⁵: Argon (18) is the previous noble gas. We then fill 4s (2), 3d (10), and 4p (5) to reach 35.
-
1s² 2s² 2p⁶: A neutral Sodium atom has 11 electrons. The Na⁺ ion loses one electron (the 3s¹ electron), leaving 10 electrons, which is isoelectronic with Neon.
-
Tellurium (Te): The 5s and 5p shells indicate the element is in the 5th period. The p⁴ configuration indicates it is the 4th element in the p-block of that period.
-
1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹ 3d⁵: Like Copper, Chromium is an exception. It moves one electron from 4s to 3d to achieve two half-filled subshells, which is more stable than [Ar] 4s² 3d⁴.
-
3 unpaired electrons: Nitrogen (1s² 2s² 2p³) has three electrons in the 2p subshell. According to Hund's Rule, these occupy the three p-orbitals individually with parallel spins.
-
1s² 2s² 2p⁶ 3s² 3p⁶: Neutral Sulfur has 16 electrons. The S²⁻ ion gains two electrons to fill its valence shell, reaching a total of 18 (isoelectronic with Argon).
-
Tin (Sn): Krypton (36) + 2 + 10 + 2 = 50. Atomic number 50 is Tin.
-
Lower Energy State: Although the 4s orbital has a higher principal quantum number than 3d, it has a lower energy level due to orbital penetration and shielding effects, making it more favorable for electrons to occupy first. This is a common concept when solving ideal gas law questions or other physical chemistry problems where energy states matter.
Quick Quiz
1. Which principle states that electrons must occupy the lowest energy orbitals first?
- A Hund's Rule
- B Pauli Exclusion Principle
- C Aufbau Principle
- D Heisenberg Uncertainty Principle
Check answer
Answer: C. Aufbau Principle
2. What is the correct electron configuration for a neutral Carbon atom (Z=6)?
- A 1s² 2s⁴
- B 1s² 2s² 2p²
- C 1s² 2p⁴
- D 1s² 2s¹ 2p³
Check answer
Answer: B. 1s² 2s² 2p²
3. How many electrons can a single 'd' subshell hold in total?
- A 2
- B 6
- C 10
- D 14
Check answer
Answer: C. 10
4. Which of these ions is isoelectronic with Argon?
- A Mg²⁺
- B O²⁻
- C Cl⁻
- D Al³⁺
Check answer
Answer: C. Cl⁻
5. Which element is an exception to the standard Aufbau filling order?
- A Iron
- B Copper
- C Zinc
- D Magnesium
Check answer
Answer: B. Copper
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the Aufbau Principle?
The Aufbau Principle states that in the ground state of an atom or ion, electrons fill atomic orbitals of the lowest available energy levels before occupying higher levels. This results in the most stable electron configuration for the atom.
Why does the 4s orbital fill before the 3d orbital?
The 4s orbital is at a lower energy level than the 3d orbital because electrons in the 4s orbital spend more time closer to the nucleus on average. This penetration effect reduces the energy of the 4s orbital below that of the 3d orbital in neutral atoms.
What does it mean for two species to be isoelectronic?
Isoelectronic species are atoms, ions, or molecules that have the same number of electrons and the same electron configuration. For example, the Na⁺ ion and the Neon atom both have 10 electrons and the configuration 1s² 2s² 2p⁶.
How do you apply Hund's Rule?
Hund's Rule is applied by placing one electron in each orbital of a subshell (like the three p-orbitals or five d-orbitals) before any pairing occurs. All electrons in these singly occupied orbitals must have the same spin to minimize electron-electron repulsion.
What are the exceptions to the electron configuration rules?
The most common exceptions occur in the transition metals, specifically Chromium (Cr) and Copper (Cu). These elements shift an electron from the s-orbital to the d-orbital to achieve a half-filled or fully-filled d-subshell, which provides extra stability. Similar logic applies to calculations found in cell potential calculations where stable states influence voltage.
How many electrons can each orbital type hold?
The s-subshell can hold 2 electrons, the p-subshell can hold 6, the d-subshell can hold 10, and the f-subshell can hold 14. Each individual orbital within these subshells can hold a maximum of 2 electrons with opposite spins.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.