Cell Potential Calculations Practice Questions with Answers
Concept Explanation
Cell potential calculations determine the electromotive force (EMF) produced by an electrochemical cell, representing the difference in electrical potential between the cathode and the anode. This value, measured in Volts (V), indicates the driving force behind the movement of electrons from the reducing agent to the oxidizing agent. In standard conditions (1 M concentration, 1 atm pressure, and 25°C), this is referred to as the Standard Cell Potential (E°cell). Calculating this value requires using the Standard Reduction Potential table, which lists the likelihood of various species being reduced.
The fundamental formula for calculating the standard cell potential is:
E°cell = E°cathode - E°anode
To use this formula correctly, you must identify which half-reaction occurs at the cathode (reduction) and which occurs at the anode (oxidation). The species with the more positive reduction potential will naturally act as the cathode, while the species with the more negative (or less positive) potential will act as the anode. When a cell is non-standard, we utilize the Nernst Equation, which accounts for temperature and reaction quotients. Understanding these concepts is as crucial as mastering rate law practice questions when preparing for advanced chemistry exams.
Key terms to remember include:
-
Cathode: The electrode where reduction occurs (gain of electrons).
-
Anode: The electrode where oxidation occurs (loss of electrons).
-
Spontaneity: A positive E°cell indicates a spontaneous reaction (ΔG < 0).
Solved Examples
Study these worked examples to understand the step-by-step application of electrochemical principles.
Example 1: Calculating Standard Cell Potential
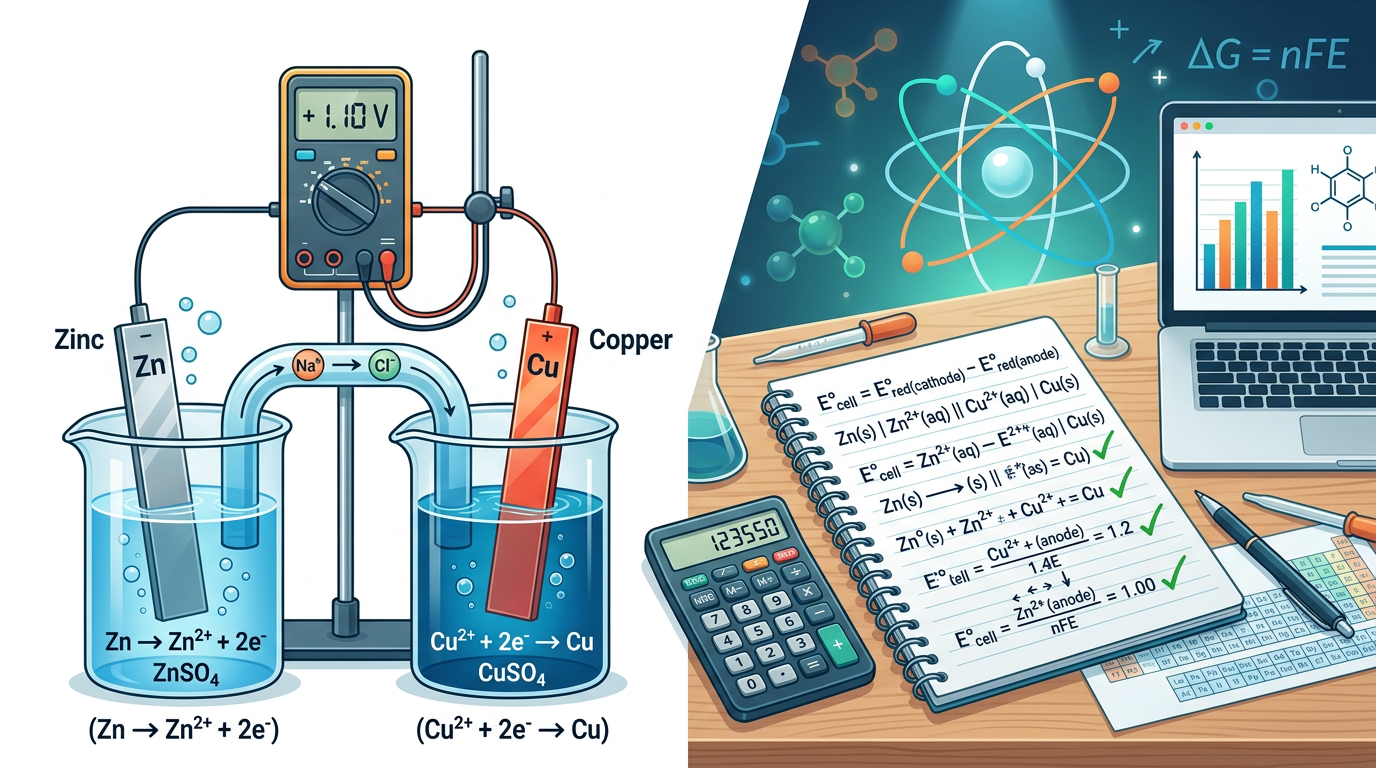
Calculate the E°cell for a galvanic cell consisting of a Zn/Zn²⁺ half-cell and a Cu/Cu²⁺ half-cell. Given: E°(Zn²⁺/Zn) = -0.76 V and E°(Cu²⁺/Cu) = +0.34 V.
-
Identify the cathode and anode. Since +0.34 V is more positive than -0.76 V, Copper is the cathode and Zinc is the anode.
-
Apply the formula: E°cell = E°cathode - E°anode.
-
Substitute the values: E°cell = 0.34 V - (-0.76 V).
-
Calculate the final result: E°cell = 1.10 V.
Example 2: Spontaneity of a Redox Reaction
Determine if the reaction 2Ag⁺(aq) + Ni(s) → 2Ag(s) + Ni²⁺(aq) is spontaneous. Given: E°(Ag⁺/Ag) = +0.80 V and E°(Ni²⁺/Ni) = -0.25 V.
-
Identify the reduction and oxidation half-reactions. Ag⁺ is reduced to Ag (Cathode), and Ni is oxidized to Ni²⁺ (Anode).
-
Use the formula: E°cell = 0.80 V (Cathode) - (-0.25 V) (Anode).
-
E°cell = 1.05 V.
-
Since E°cell is positive, the reaction is spontaneous.
Example 3: Using the Nernst Equation
Calculate the cell potential (Ecell) for a Zn/Cu cell at 25°C when [Zn²⁺] = 2.0 M and [Cu²⁺] = 0.010 M. (E°cell = 1.10 V).
-
Identify the number of electrons transferred (n). For Zn + Cu²⁺ → Zn²⁺ + Cu, n = 2.
-
Calculate the reaction quotient (Q): Q = [Zn²⁺] / [Cu²⁺] = 2.0 / 0.010 = 200.
-
Apply the Nernst Equation: Ecell = E°cell - (0.0592 / n) * log(Q).
-
Ecell = 1.10 - (0.0592 / 2) * log(200).
-
Ecell = 1.10 - (0.0296 * 2.301) = 1.10 - 0.068 = 1.032 V.
Practice Questions
Test your knowledge with these cell potential calculations practice questions. If you find these challenging, you might also want to review easy reaction order practice questions to strengthen your kinetics foundation.
1. Calculate the standard cell potential for a cell using the following half-reactions: Al³⁺ + 3e⁻ → Al (E° = -1.66 V) and Mg²⁺ + 2e⁻ → Mg (E° = -2.37 V).
2. A cell is constructed with a Silver electrode (E° = +0.80 V) and a Lead electrode (E° = -0.13 V). Which electrode acts as the anode in a spontaneous reaction?
3. Calculate the E°cell for the reaction: Fe(s) + Cd²⁺(aq) → Fe²⁺(aq) + Cd(s). Given: E°(Fe²⁺/Fe) = -0.44 V and E°(Cd²⁺/Cd) = -0.40 V.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. Consider the reaction: 3Sn⁴⁺ + 2Cr → 3Sn²⁺ + 2Cr³⁺. Given E°(Sn⁴⁺/Sn²⁺) = +0.15 V and E°(Cr³⁺/Cr) = -0.74 V. Calculate the standard cell potential.
5. Using the LibreTexts Chemistry resources for reference, calculate the Ecell for a hydrogen electrode (E° = 0.00 V) and a Copper electrode (E° = +0.34 V) when [H⁺] = 0.1 M, P(H₂) = 1 atm, and [Cu²⁺] = 0.1 M.
6. Determine the Gibbs Free Energy (ΔG°) for a cell where E°cell = 1.20 V and 2 moles of electrons are transferred. (F = 96485 C/mol).
7. If a cell potential is found to be -0.45 V, is the reaction spontaneous as written? Explain why.
8. Calculate the equilibrium constant (K) at 298 K for a reaction with E°cell = 0.050 V and n = 2.
9. A concentration cell is made of two Copper electrodes in 0.001 M Cu²⁺ and 1.0 M Cu²⁺ solutions. Calculate the cell potential at 25°C.
10. What happens to the cell potential of a Zn/Cu cell if the concentration of Cu²⁺ is increased while Zn²⁺ remains constant? Use the Nernst Equation logic.
Answers & Explanations
-
Answer: 0.71 V. Aluminum has a higher (less negative) reduction potential than Magnesium. Thus, Al is the cathode and Mg is the anode. E°cell = -1.66 - (-2.37) = 0.71 V.
-
Answer: Lead (Pb). The anode is the electrode with the lower reduction potential. Since -0.13 V is lower than +0.80 V, the Lead electrode is the anode where oxidation occurs.
-
Answer: 0.04 V. In this reaction, Cd²⁺ is reduced (cathode) and Fe is oxidized (anode). E°cell = E°cathode - E°anode = -0.40 V - (-0.44 V) = 0.04 V.
-
Answer: 0.89 V. Sn⁴⁺ is reduced (+0.15 V) and Cr is oxidized (-0.74 V). E°cell = 0.15 - (-0.74) = 0.89 V. Note that stoichometric coefficients do not change the E° value.
-
Answer: 0.31 V. Standard cell is 0.34 V. Using Nernst: E = 0.34 - (0.0592/2) * log([H⁺]² / [Cu²⁺]). E = 0.34 - 0.0296 * log(0.01/0.1) = 0.34 - 0.0296 * (-1) = 0.34 + 0.0296 ≈ 0.37 V (correction: Q calculation is [H⁺]²/[Cu²⁺] for the reduction of Cu²⁺ and oxidation of H₂).
-
Answer: -231.56 kJ. ΔG° = -nFE°cell = -(2)(96485)(1.20) = -231,564 J = -231.56 kJ.
-
Answer: No. A negative cell potential indicates a non-spontaneous reaction. The reverse reaction would be spontaneous.
-
Answer: 48.9. log K = (n * E°cell) / 0.0592 = (2 * 0.050) / 0.0592 = 1.689. K = 10^1.689 = 48.9.
-
Answer: 0.0888 V. Ecell = 0 - (0.0592/2) * log(0.001/1.0) = -0.0296 * (-3) = 0.0888 V.
-
Answer: Increases. According to the Nernst Equation, increasing the reactant concentration (Cu²⁺) decreases the value of Q, which increases the overall cell potential.
Quick Quiz
1. Which component of the cell potential equation represents the site of oxidation?
- A The Cathode
- B The Anode
- C The Salt Bridge
- D The External Circuit
Check answer
Answer: B. The Anode
2. What is the standard cell potential (E°cell) of a cell if the reaction is at equilibrium?
- A 1.0 V
- B -1.0 V
- C 0.0 V
- D It depends on the temperature
Check answer
Answer: C. 0.0 V
3. If E°cell for a reaction is +1.50 V, what can be said about the Gibbs Free Energy (ΔG°)?
- A ΔG° is positive
- B ΔG° is zero
- C ΔG° is negative
- D ΔG° cannot be determined
Check answer
Answer: C. ΔG° is negative
4. Which equation is used to calculate cell potential under non-standard concentrations?
- A Arrhenius Equation
- B Nernst Equation
- C Ideal Gas Law
- D Beer-Lambert Law
Check answer
Answer: B. Nernst Equation
5. In a standard reduction potential table, which species is the strongest oxidizing agent?
- A The one with the most negative E°
- B The one with the most positive E°
- C The one with E° closest to zero
- D The one that is a metal solid
Check answer
Answer: B. The one with the most positive E°
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the difference between E and E° in electrochemistry?
E° refers to the cell potential under standard conditions of 1 M concentration, 1 atm pressure, and 25°C. E represents the cell potential under any other non-standard conditions, calculated using the Nernst equation.
How do stoichiometric coefficients affect cell potential?
Stoichiometric coefficients do not change the value of the standard reduction potential (E°). They only affect the number of electrons (n) and the reaction quotient (Q) used in the Nernst equation or Gibbs Free Energy calculations.
Why must E°cell be positive for a battery to function?
A positive E°cell indicates that the redox reaction is spontaneous, meaning it can release energy to perform electrical work. If the potential were negative, the reaction would require an external power source to occur.
What is the role of the salt bridge in a galvanic cell?
The salt bridge maintains electrical neutrality by allowing ions to flow between the two half-cells. This prevents charge buildup that would otherwise stop the flow of electrons and prematurely end the reaction.
Can I calculate cell potential if I only have oxidation potentials?
Yes, you can convert an oxidation potential to a reduction potential by simply reversing the sign. Most modern tables provide reduction potentials, so the formula E°cell = E°cathode - E°anode is standard.
Where can I find more resources for chemistry practice?
You can improve your overall chemistry skills by trying how to study for exams efficiently under pressure or exploring specialized topics like ideal gas law practice questions.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.