Easy Molality Practice Questions
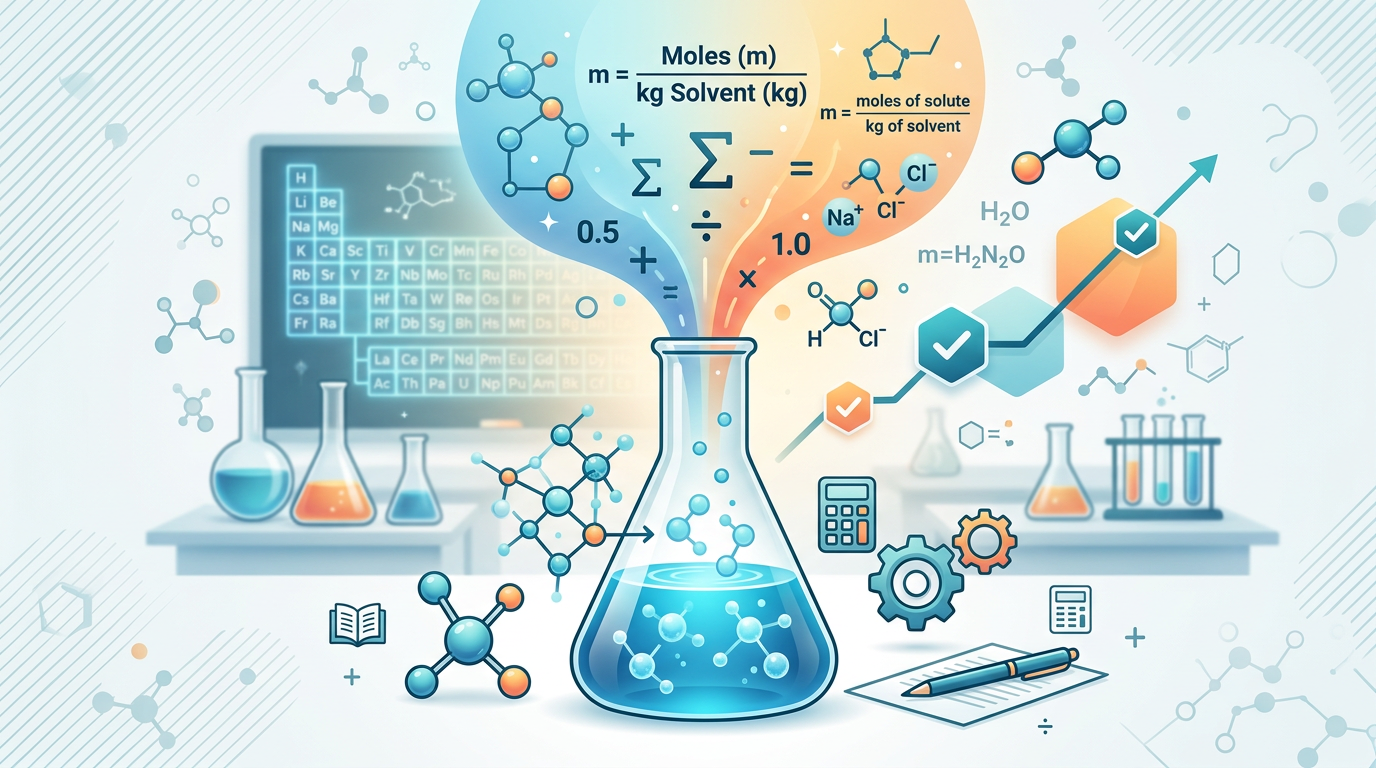
Concept Explanation
Molality is a measure of concentration that expresses the number of moles of solute dissolved in exactly one kilogram (1 kg) of solvent. Unlike molarity, which depends on the volume of the solution, molality is based solely on mass, making it independent of temperature and pressure changes. This property makes it particularly useful in physical chemistry, especially when calculating colligative properties like boiling point elevation or freezing point depression. To calculate molality, you use the formula: molality (m) = moles of solute / mass of solvent (kg). If you are starting with mass in grams, you will first need to perform grams to moles conversions for the solute and convert the solvent's mass from grams to kilograms.
According to the IUPAC Gold Book, the standard unit for molality is mol/kg, often denoted by a lowercase 'm'. When working through these easy molality practice questions, always ensure your solvent mass is in kilograms. If a problem provides the total mass of the solution, you must subtract the mass of the solute to find the mass of the solvent alone. Understanding the molarity vs molality difference is key to choosing the right concentration unit for your specific laboratory application.
Solved Examples
Review these step-by-step solutions to understand the basic mechanics of molality calculations.
-
Example 1: Basic Molality Calculation
Calculate the molality of a solution where 0.5 moles of sodium chloride (NaCl) are dissolved in 2.0 kg of water.-
Identify the moles of solute: 0.5 mol.
-
Identify the mass of solvent in kg: 2.0 kg.
-
Apply the formula: m = 0.5 mol / 2.0 kg.
-
Solve: m = 0.25 m.
-
-
Example 2: Converting Solvent Grams to Kilograms
What is the molality of a solution containing 2.0 moles of glucose in 500 grams of water?-
Identify moles of solute: 2.0 mol.
-
Convert solvent mass to kg: 500 g / 1000 = 0.5 kg.
-
Apply the formula: m = 2.0 mol / 0.5 kg.
-
Solve: m = 4.0 m.
-
-
Example 3: Starting from Solute Mass
Determine the molality of a solution made by dissolving 40.0 g of NaOH (molar mass = 40.0 g/mol) in 1.0 kg of water.-
Convert mass of solute to moles: 40.0 g / 40.0 g/mol = 1.0 mole.
-
Identify mass of solvent: 1.0 kg.
-
Apply the formula: m = 1.0 mol / 1.0 kg.
-
Solve: m = 1.0 m.
-
Practice Questions
Test your skills with these easy molality practice questions. Remember to keep track of your units!
-
A solution is prepared by dissolving 0.30 moles of sucrose in 0.60 kg of water. What is the molality?
-
Calculate the molality of 1.5 moles of KNO₃ dissolved in 750 grams of water.
-
If you dissolve 0.10 moles of silver nitrate in 200 grams of water, what is the resulting molality?
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →-
How many moles of solute are needed to create a 2.0 m solution using 3.0 kg of solvent?
-
Calculate the molality of a solution containing 58.5 g of NaCl (molar mass = 58.5 g/mol) in 2.0 kg of water.
-
What is the molality of 0.45 moles of urea dissolved in 1500 g of water?
-
A chemist dissolves 10.0 g of HF (molar mass = 20.0 g/mol) in 500 g of water. Find the molality.
-
How many kilograms of water are required to dissolve 0.8 moles of a solute to make a 0.4 m solution?
-
Calculate the molality of 2.5 moles of ethanol in 4.0 kg of water.
-
Find the molality of a solution where 4.0 g of Helium (molar mass = 4.0 g/mol) is mixed with 250 g of water.
Answers & Explanations
Check your work against the detailed explanations provided below.
-
Answer: 0.50 m. Use the formula: 0.30 mol / 0.60 kg = 0.50 mol/kg.
-
Answer: 2.0 m. First, convert 750 g to 0.75 kg. Then, 1.5 moles / 0.75 kg = 2.0 m.
-
Answer: 0.50 m. Convert 200 g to 0.20 kg. Then, 0.10 moles / 0.20 kg = 0.50 m.
-
Answer: 6.0 moles. Rearrange the formula: Moles = molality × kg solvent. 2.0 m × 3.0 kg = 6.0 moles.
-
Answer: 0.50 m. First, convert grams of NaCl to moles: 58.5 g / 58.5 g/mol = 1.0 mole. Then, 1.0 mole / 2.0 kg = 0.50 m.
-
Answer: 0.30 m. Convert 1500 g to 1.5 kg. Then, 0.45 moles / 1.5 kg = 0.30 m.
-
Answer: 1.0 m. First, find moles of HF: 10.0 g / 20.0 g/mol = 0.5 moles. Convert 500 g to 0.5 kg. 0.5 mol / 0.5 kg = 1.0 m.
-
Answer: 2.0 kg. Rearrange the formula: kg solvent = moles / molality. 0.8 mol / 0.4 m = 2.0 kg.
-
Answer: 0.625 m. 2.5 moles / 4.0 kg = 0.625 m.
-
Answer: 4.0 m. First, find moles of He: 4.0 g / 4.0 g/mol = 1.0 mole. Convert 250 g to 0.25 kg. 1.0 mol / 0.25 kg = 4.0 m.
Quick Quiz
1. Which of the following is the correct formula for molality?
- A Moles of solute / Liters of solution
- B Grams of solute / Kilograms of solvent
- C Moles of solute / Kilograms of solvent
- D Moles of solute / Grams of solution
Check answer
Answer: C. Moles of solute / Kilograms of solvent
2. Why is molality used instead of molarity in experiments involving temperature changes?
- A Molality is easier to calculate.
- B Mass does not change with temperature, but volume does.
- C Molality uses moles instead of grams.
- D Molarity is only for gases.
Check answer
Answer: B. Mass does not change with temperature, but volume does.
3. If you have 2 moles of solute in 2000 grams of solvent, what is the molality?
- A 1.0 m
- B 2.0 m
- C 0.5 m
- D 4.0 m
Check answer
Answer: A. 1.0 m
4. What is the standard unit for molality?
- A M
- B mol/L
- C mol/kg
- D g/mL
Check answer
Answer: C. mol/kg
5. To calculate molality, what must you do if the solvent mass is given in grams?
- A Multiply by 1000
- B Divide by 1000
- C Add the solute mass
- D Subtract the solute mass
Check answer
Answer: B. Divide by 1000
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the difference between molarity and molality?
Molarity measures moles of solute per liter of total solution, while molality measures moles of solute per kilogram of solvent. Because volume expands or contracts with temperature, molarity changes with heat, whereas molality remains constant.
Can molality ever be equal to molarity?
Molality and molarity are approximately equal in very dilute aqueous solutions at room temperature. This occurs because the density of water is roughly 1.0 g/mL, meaning 1 kg of solvent is nearly equal to 1 L of solution when the solute volume is negligible.
Why does molality use mass of solvent instead of mass of solution?
Using the mass of the solvent ensures that the concentration ratio remains fixed regardless of the amount of solute added. This consistency is required for calculating properties like boiling point elevation, which depend on the ratio of solute particles to solvent molecules.
Is molality affected by pressure?
No, molality is not affected by pressure. Since mass is invariant under pressure changes, molality provides a stable concentration measurement for high-pressure chemical reactions where volume might fluctuate.
How do you convert grams of solute to moles for molality?
To convert grams to moles, divide the mass of the solute by its molar mass. You can find the molar mass by summing the atomic weights of the elements in the chemical formula using a periodic table.
What is the symbol for molality?
The symbol for molality is a lowercase italicized "m". It is important not to confuse this with the uppercase "M" used for molarity or the "m" used for mass in physics equations.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.
Related Articles

Hard MCAT Carbonyl Practice Questions
Master high-yield MCAT carbonyl chemistry with our hard practice questions, detailed explanations, and expert strategies for nucleophilic reactions.
May 11, 2026

Hard MCAT Organic Reactions Practice Questions
Hard MCAT Organic Reactions Practice Questions
May 11, 2026

Hard MCAT Nomenclature Practice Questions
Hard MCAT Nomenclature Practice Questions
May 11, 2026