Hard SN1 vs SN2 Reaction Practice Questions
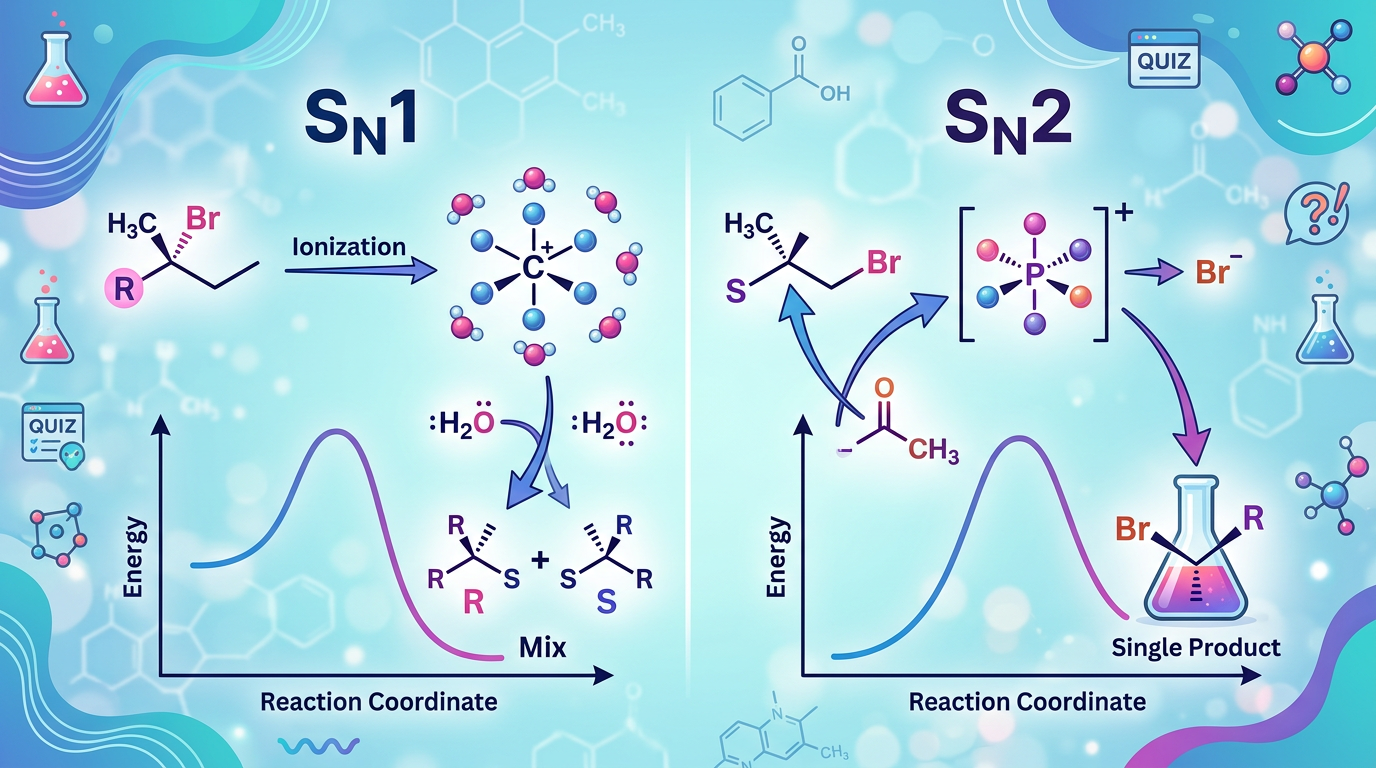
Concept Explanation
The distinction between SN1 and SN2 pathways depends on whether a nucleophilic substitution occurs through a two-step mechanism involving a carbocation intermediate (SN1) or a single-step concerted process (SN2). In an SN1 (Substitution Nucleophilic Unimolecular) reaction, the rate-determining step involves only the substrate, leading to a planar carbocation that can be attacked from either side, typically resulting in racemization. Conversely, the SN2 (Substitution Nucleophilic Bimolecular) reaction involves a simultaneous attack by the nucleophile and departure of the leaving group, resulting in a characteristic inversion of stereochemical configuration, often referred to as Walden inversion. Understanding these mechanisms is a cornerstone of organic chemistry, much like mastering reaction mechanism practice questions in other advanced topics.
Several factors dictate which pathway a reaction will follow:
Factor SN1 Preference SN2 Preference Substrate Structure Tertiary (3°) > Secondary (2°) Methyl > Primary (1°) > Secondary (2°) Nucleophile Weak (e.g., H2O, ROH) Strong (e.g., OH-, CN-, I-) Solvent Polar Protic (stabilizes ions) Polar Aprotic (enhances nucleophile) Stereochemistry Racemization Inversion
According to IUPAC definitions, the unimolecularity or bimolecularity refers specifically to the molecularity of the rate-determining step. In competitive "hard" scenarios, such as secondary substrates, the choice of solvent and the strength of the nucleophile become the deciding factors. For more on how molecular shapes influence reactivity, you might explore VSEPR geometry practice questions.
Solved Examples
-
Predict the product and mechanism for (S)-2-bromobutane reacting with Sodium Cyanide (NaCN) in DMSO.
-
Identify the substrate: 2-bromobutane is a secondary (2°) alkyl halide.
-
Identify the nucleophile: CN- is a strong, small nucleophile.
-
Identify the solvent: DMSO is a polar aprotic solvent, which favors SN2.
-
Determine mechanism: The combination of a strong nucleophile and polar aprotic solvent on a 2° substrate points to SN2.
-
Determine stereochemistry: SN2 causes inversion. (S) becomes (R).
-
Final Answer: (R)-2-cyanobutane via SN2.
-
-
Analyze the reaction of 1-bromo-1-methylcyclohexane with methanol (CH3OH) at room temperature.
-
Identify the substrate: The carbon holding the Br is attached to three other carbons (tertiary).
-
Identify the nucleophile: Methanol is a weak nucleophile and also the solvent (solvolysis).
-
Identify the solvent: Methanol is polar protic.
-
Determine mechanism: Tertiary substrates cannot undergo SN2 due to steric hindrance; therefore, SN1 is the only viable substitution pathway.
-
Final Answer: 1-methoxy-1-methylcyclohexane via SN1.
-
-
Compare the reaction rates of 1-chlorobutane and 1-chloro-2,2-dimethylpropane with NaI in acetone.
-
Identify conditions: NaI in acetone is a classic SN2 environment (strong nucleophile, polar aprotic solvent).
-
Compare steric hindrance: 1-chlorobutane is a primary halide. 1-chloro-2,2-dimethylpropane (neopentyl chloride) is also primary but has a massive tert-butyl group adjacent to the reaction center.
-
Evaluate: Neopentyl systems are notoriously slow in SN2 because the bulky groups block the backside attack.
-
Final Answer: 1-chlorobutane reacts significantly faster.
-
Practice Questions
1. Which mechanism is favored when (R)-3-chloro-3-methylhexane is heated in ethanol, and what is the stereochemical outcome?
2. Rank the following in order of increasing SN2 reactivity: 2-bromo-2-methylbutane, 1-bromopentane, 2-bromopentane.
3. A secondary alkyl tosylate is reacted with Sodium Azide (NaN3) in DMF. If the starting material was enantiopure, will the product be a racemate or a single enantiomer?
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. Explain why 1-bromobicyclo[2.2.1]heptane is virtually inert toward both SN1 and SN2 conditions.
5. Predict the major substitution product when (3S,4S)-3-iodohexane reacts with Sodium Acetate in DMSO.
6. How does the addition of silver nitrate (AgNO3) in water affect the rate and mechanism of the substitution of 2-chloro-2-methylpropane?
7. Consider the reaction of methyl iodide with hydroxide in a mixture of water and ethanol. If the concentration of methyl iodide is doubled, what happens to the rate?
8. Why does 3-bromocyclohexene react much faster than bromocyclohexane in an SN1 reaction using aqueous acetone?
9. Identify the better leaving group between a triflate (-OTf) and a mesylate (-OMs) and explain its impact on SN2 reaction kinetics.
10. Predict the effect on the SN2 reaction rate if the solvent is changed from Methanol to Acetonitrile for the reaction of ethyl bromide with potassium cyanide.
Answers & Explanations
-
SN1; Racemization. The substrate is tertiary (3°), which prevents SN2. Ethanol is a weak nucleophile and polar protic solvent, favoring the formation of a carbocation. The resulting planar intermediate allows attack from both faces.
-
2-bromo-2-methylbutane < 2-bromopentane < 1-bromopentane. SN2 reactivity is governed by steric hindrance. Tertiary (3°) is the slowest (virtually zero), followed by secondary (2°), with primary (1°) being the fastest.
-
Single Enantiomer (Inversion). Sodium Azide is a strong nucleophile and DMF is a polar aprotic solvent. These conditions strongly favor SN2 on a secondary substrate, leading to 100% inversion of configuration.
-
Bridgehead Constraint. In SN2, the nucleophile cannot attack from the backside because it is blocked by the cage structure. In SN1, the resulting carbocation cannot achieve the required planar geometry (Bredt's Rule violation/strain), making the intermediate too high in energy to form. Reference: Bredt's Rule.
-
(3R,4S)-3-acetoxyhexane. The reaction is SN2 due to the strong nucleophile (acetate) and polar aprotic solvent (DMSO). Only the stereocenter at C3 undergoes inversion, changing from S to R.
-
Increases rate; favors SN1. Silver ions coordinate with the leaving group (Cl-), facilitating its departure to form a carbocation. This accelerates the rate-determining step of the SN1 mechanism.
-
Rate doubles. The reaction of methyl iodide (primary) with hydroxide is an SN2 process. The rate law is Rate = k[Substrate][Nucleophile]. Doubling the substrate concentration directly doubles the rate.
-
Resonance Stabilization. 3-bromocyclohexene forms an allylic carbocation upon the loss of the leaving group. This carbocation is resonance-stabilized, lowering the activation energy for the SN1 pathway compared to the secondary carbocation from bromocyclohexane.
-
Triflate is better. The triflate ion is a much weaker base due to the strong electron-withdrawing effect of the three fluorine atoms. A better leaving group lowers the transition state energy, significantly increasing the SN2 reaction rate.
-
Rate Increases. Acetonitrile is polar aprotic, while methanol is polar protic. Protic solvents solvate the nucleophile (CN-) through hydrogen bonding, creating a "solvent shell" that must be broken. Aprotic solvents leave the nucleophile "naked" and more reactive.
Quick Quiz
1. Which of the following solvents would most likely accelerate an SN2 reaction between CH3I and NaN3?
- A Water
- B Ethanol
- C Dimethylsulfoxide (DMSO)
- D Acetic Acid
Check answer
Answer: C. Dimethylsulfoxide (DMSO)
2. What is the primary reason tertiary alkyl halides do not undergo SN2 reactions?
- A The carbocation is too stable
- B Steric hindrance prevents the nucleophile from reaching the alpha-carbon
- C The leaving group is too bulky
- D The transition state is too low in energy
Check answer
Answer: B. Steric hindrance prevents the nucleophile from reaching the alpha-carbon
3. Which substrate will react fastest in an SN1 reaction?
- A Methyl bromide
- B Ethyl bromide
- C Isopropyl bromide
- D tert-Butyl bromide
Check answer
Answer: D. tert-Butyl bromide
4. If an SN2 reaction is performed on a chiral starting material with an (R) configuration, the product will:
- A Always be (R)
- B Always be (S)
- C Be a 50/50 mixture of (R) and (S)
- D Depend on the priority of the nucleophile relative to the leaving group
Check answer
Answer: B. Always be (S)
5. In an SN1 reaction, doubling the concentration of the nucleophile will:
- A Double the rate
- B Quadruple the rate
- C Have no effect on the rate
- D Halve the rate
Check answer
Answer: C. Have no effect on the rate
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the main difference between SN1 and SN2?
The main difference is the timing of the mechanism; SN1 is a two-step process involving a carbocation intermediate, while SN2 is a single-step concerted process. Consequently, SN1 rate depends only on the substrate, whereas SN2 rate depends on both the substrate and the nucleophile.
Can a primary alkyl halide ever undergo an SN1 reaction?
Primary alkyl halides generally do not undergo SN1 because primary carbocations are highly unstable. However, if the carbocation can be resonance-stabilized, such as in benzylic or allylic positions, a primary substrate may follow the SN1 pathway.
Why are polar aprotic solvents preferred for SN2?
Polar aprotic solvents do not have hydrogen atoms bonded to electronegative elements, meaning they cannot hydrogen-bond with the nucleophile. This leaves the nucleophile less solvated and more energetic, which significantly increases the rate of the SN2 attack. For more on molecular properties, see our hard polarity determination practice questions.
What is the role of the leaving group in these reactions?
The leaving group must be able to stabilize the negative charge it carries away; therefore, weak bases make the best leaving groups. In both SN1 and SN2, a better leaving group lowers the activation energy and increases the overall reaction rate.
Does temperature affect the competition between SN1 and SN2?
While temperature affects all reaction rates, it more significantly influences the competition between substitution and elimination (E1/E2). Higher temperatures generally favor elimination pathways over substitution pathways due to the increase in entropy associated with forming more molecules.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.