Hard Le Chatelier’s Principle Practice Questions
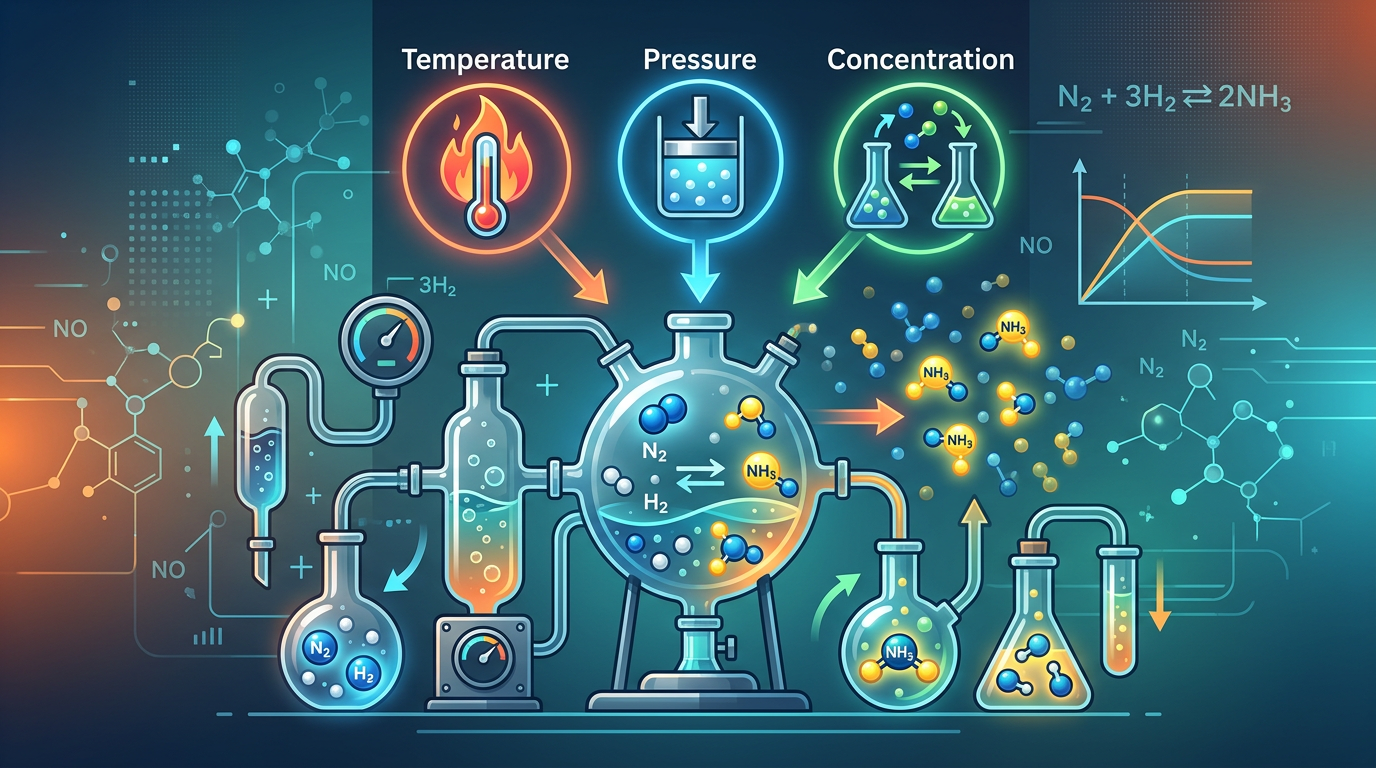
Concept Explanation
Le Chatelier’s Principle states that if a system at equilibrium is subjected to a change in concentration, temperature, volume, or pressure, the system will shift its equilibrium position to counteract the effect of that disturbance. This fundamental rule of chemical kinetics and thermodynamics allows chemists to predict how a reaction will respond to external stressors. For example, according to the Wikipedia entry on Le Chatelier's principle, the rule is widely applied in industrial processes like the Haber process to maximize the yield of ammonia. Key factors include the addition or removal of reactants/products, changes in total pressure (for gaseous systems), and temperature fluctuations which depend on whether the reaction is exothermic or endothermic. Understanding these shifts often requires a deep dive into enthalpy changes to determine how heat behaves as a reactant or product.
Key Factors Influencing Equilibrium
-
Concentration: Adding a substance shifts the equilibrium away from that substance; removing it shifts the equilibrium toward it.
-
Pressure/Volume: Increasing pressure (decreasing volume) shifts the equilibrium toward the side with fewer moles of gas.
-
Temperature: Increasing temperature shifts the equilibrium in the direction of the endothermic reaction (absorbing the extra heat).
-
Catalysts: These do NOT shift the equilibrium position; they only allow the system to reach equilibrium faster by lowering activation energy.
Solved Examples
Study these complex scenarios to understand how multiple variables interact in a chemical system.
-
Example 1: The Haber Process
Consider the reaction: N₂(g) + 3H₂(g) ⇌ 2NH₃(g) ΔH = -92 kJ/mol. What happens if the volume is halved and the temperature is increased?-
Analyze Pressure: Halving the volume doubles the pressure. The system shifts to the side with fewer gas moles (the right/product side).
-
Analyze Temperature: Since the reaction is exothermic (negative ΔH), heat is a product. Increasing temperature shifts the equilibrium to the left (reactant side).
-
Net Result: The two effects oppose each other. The final yield depends on the magnitude of each change, but typically, high pressure and moderate temperatures are used industrially to balance rate and yield.
-
-
Example 2: Complex Ion Formation
Reaction: [Co(H₂O)₆]²⁺(aq) + 4Cl⁻(aq) ⇌ [CoCl₄]²⁻(aq) + 6H₂O(l) (Pink to Blue). If concentrated HCl is added and the solution is placed in an ice bath, what color change occurs?-
Concentration: Adding HCl increases Cl⁻ concentration, shifting the equilibrium to the right (turning blue).
-
Temperature: This reaction is endothermic. Placing it in an ice bath (removing heat) shifts it to the left (turning pink).
-
Integration: The concentration shift promotes blue, while the cooling promotes pink.
-
-
Example 3: Partial Pressure and Inert Gas
In a rigid container, the reaction 2SO₂(g) + O₂(g) ⇌ 2SO₃(g) is at equilibrium. If Helium gas is added at constant volume, what happens to the yield of SO₃?-
Identify the stress: Adding an inert gas at constant volume increases total pressure but does NOT change the partial pressures of the reacting gases.
-
Conclusion: Because the concentrations (moles/volume) of the reactants and products remain unchanged, there is no shift in equilibrium.
-
Practice Questions
-
The decomposition of calcium carbonate is endothermic: CaCO₃(s) ⇌ CaO(s) + CO₂(g). If the container volume is decreased at constant temperature, how does the mass of CaCO₃ change?
-
For the reaction PCl₅(g) ⇌ PCl₃(g) + Cl₂(g) ΔH = +88 kJ/mol, list three specific actions that would maximize the concentration of PCl₅.
-
Consider the equilibrium: 2NO(g) + Br₂(g) ⇌ 2NOBr(g) ΔH = -30 kJ/mol. If the temperature is decreased and NOBr is simultaneously removed, in which direction will the equilibrium shift?
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →-
In the system N₂O₄(g) ⇌ 2NO₂(g), the forward reaction is endothermic. If the total pressure is increased by adding N₂O₄ gas, describe the immediate and long-term changes to the concentration of NO₂.
-
A saturated solution of AgCl is in equilibrium: AgCl(s) ⇌ Ag⁺(aq) + Cl⁻(aq). If AgNO₃ is added, what happens to the solubility of AgCl? (Hint: This relates to equilibrium constants in aqueous solutions).
-
The reaction 2HI(g) ⇌ H₂(g) + I₂(g) is exothermic. If the volume of the container is doubled, what is the effect on the equilibrium constant (K) and the position of equilibrium?
-
For the gas-phase reaction A + B ⇌ C + D, ΔH is unknown. When the temperature is increased, the concentration of C decreases. Is the forward reaction exothermic or endothermic?
-
In a closed system containing CO(g) + H₂O(g) ⇌ CO₂(g) + H₂(g), how does adding a catalyst affect the final pressure of the system?
-
Explain why changing the volume of a reaction vessel has no effect on the equilibrium position for the reaction: H₂(g) + Cl₂(g) ⇌ 2HCl(g).
-
A mixture of SO₂, O₂, and SO₃ is at equilibrium. If the temperature is increased and the color of the mixture darkens (assume SO₃ is colorless and SO₂/O₂ have a slight tint), what can be concluded about the enthalpy of the forward reaction?
Answers & Explanations
-
Answer: Mass of CaCO₃ increases. Decreasing volume increases the partial pressure of CO₂ gas. To counteract this, the system shifts to the left (toward the solid side) to consume CO₂ gas, thus producing more solid CaCO₃.
-
Answer: Decrease temperature, increase pressure, add PCl₃ or Cl₂. Since the reaction is endothermic, lowering heat shifts it left. Increasing pressure shifts it toward the side with fewer gas moles (the reactant side). Adding products also drives the reaction backward.
-
Answer: Shift to the right (Product side). Decreasing temperature favors the exothermic (forward) direction. Removing a product (NOBr) also pulls the equilibrium toward the right. Both actions work in the same direction.
-
Answer: Immediate increase, then a partial decrease. Adding N₂O₄ immediately increases its concentration. According to Le Chatelier, the system shifts right to consume the extra N₂O₄, increasing NO₂ concentration further until a new equilibrium is reached.
-
Answer: Solubility decreases. Adding AgNO₃ introduces Ag⁺ ions (common ion effect). This shifts the equilibrium to the left, causing more AgCl(s) to precipitate, thus reducing its solubility.
-
Answer: K remains constant; no shift in equilibrium. The equilibrium constant K only changes with temperature. Since there are 2 moles of gas on both sides, changing volume/pressure does not affect the position of equilibrium.
-
Answer: Exothermic. If increasing temperature decreases the product concentration (C), the system shifted toward the reactants. This happens when heat is a product, meaning the forward reaction is exothermic.
-
Answer: No effect. A catalyst increases the rate of both forward and reverse reactions equally. It does not change the equilibrium concentrations or the total pressure of the system.
-
Answer: Equal moles of gas. There are 2 moles of gaseous reactants (1 H₂ + 1 Cl₂) and 2 moles of gaseous product (2 HCl). Pressure/volume changes only affect systems where there is a change in the number of gas moles.
-
Answer: The forward reaction is exothermic. Increasing temperature shifts the equilibrium in the endothermic direction. If the color darkens (indicating more reactants), the reverse reaction is endothermic, meaning the forward reaction must be exothermic. More details on energy profiles can be found in our guide on heat of reaction practice.
Quick Quiz
1. What happens to the equilibrium constant (K) of an endothermic reaction when the temperature is increased?
- A It decreases
- B It remains the same
- C It increases
- D It becomes zero
Check answer
Answer: C. It increases
2. In the reaction 2A(g) + B(s) ⇌ 3C(g), what is the effect of increasing the container volume?
- A Shift to the left
- B Shift to the right
- C No shift occurs
- D The solid B disappears
Check answer
Answer: B. Shift to the right
3. Which of the following stressors will change the value of the equilibrium constant?
- A Adding a catalyst
- B Changing the concentration of reactants
- C Changing the volume of the vessel
- D Changing the temperature
Check answer
Answer: D. Changing the temperature
4. For the exothermic reaction A ⇌ B, how does cooling the system affect the yield of B?
- A Yield of B increases
- B Yield of B decreases
- C Yield of B remains constant
- D Yield of B fluctuates indefinitely
Check answer
Answer: A. Yield of B increases
5. Adding an inert gas to a system at constant pressure (allowing volume to change) will shift the equilibrium toward:
- A The side with fewer moles of gas
- B The side with more moles of gas
- C The side with higher enthalpy
- D The reactant side always
Check answer
Answer: B. The side with more moles of gas
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
Does a catalyst shift the equilibrium position?
No, a catalyst does not shift the equilibrium position because it speeds up the forward and reverse reactions by the same factor. It simply allows the system to reach the same equilibrium state in a shorter period of time.
What is the common ion effect in Le Chatelier’s Principle?
The common ion effect occurs when a salt containing an ion already present in the equilibrium mixture is added, causing the equilibrium to shift to consume that ion. This typically results in the precipitation of a sparingly soluble salt, as seen in LibreTexts Chemistry resources.
How does adding an inert gas affect equilibrium?
If an inert gas is added at constant volume, it has no effect on the equilibrium because the partial pressures of the reacting gases remain unchanged. However, if added at constant pressure, the volume increases, which shifts the equilibrium toward the side with more moles of gas.
Why does temperature change the equilibrium constant while concentration does not?
Temperature changes the kinetic energy of particles and the proportion of molecules that can overcome the activation energy barrier for both directions differently. Concentration changes only shift the position to maintain the same ratio defined by the constant K.
Can Le Chatelier’s Principle be applied to physical changes?
Yes, it applies to physical equilibria such as phase changes. For example, increasing pressure on ice shifts the equilibrium toward the liquid phase (water) because water is more dense than ice, occupying less volume.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.