Hard Hybridization Practice Questions
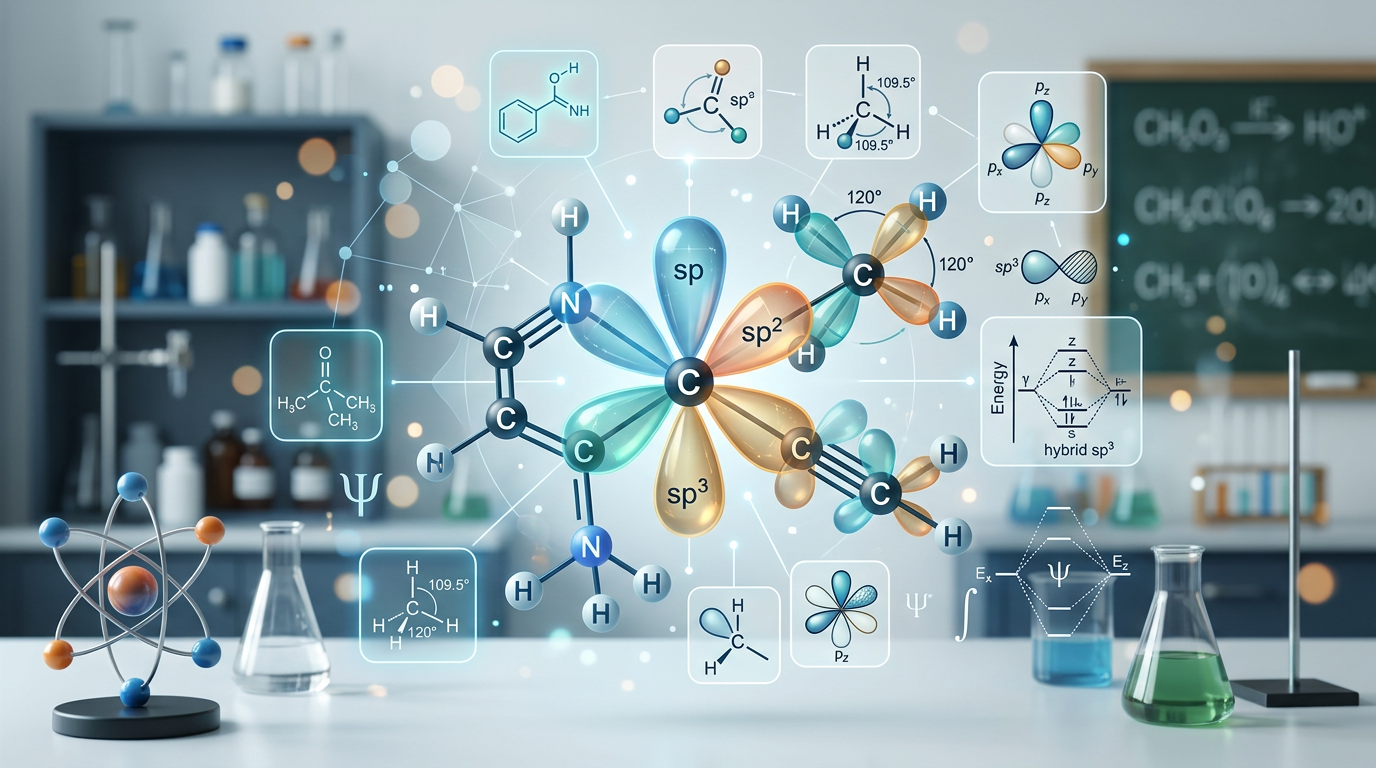
Concept Explanation
Hybridization is the mathematical process of mixing atomic orbitals, such as s, p, and d orbitals, to create new, degenerate hybrid orbitals that describe the bonding properties and geometries of polyatomic molecules. This model, central to Valence Bond Theory, explains why atoms like carbon can form four equivalent bonds in methane despite having differing energy levels in their ground-state valence electrons. By calculating the steric number (the sum of sigma bonds and lone pairs on a central atom), chemists can predict whether an atom adopts sp, sp2, sp3, sp3d, or sp3d2 hybridization. For instance, a steric number of 4 typically indicates sp3 hybridization, resulting in a tetrahedral arrangement. Understanding these advanced electronic configurations is essential for mastering topics like Lewis structures and molecular orbital theory.
In complex molecules, hybridization also accounts for the formation of multiple bonds. Sigma (σ) bonds result from the head-on overlap of hybrid orbitals, while pi (π) bonds arise from the sideways overlap of unhybridized p-orbitals. This distinction is vital when analyzing molecules with resonance or expanded octets, where d-orbitals may participate in the hybrid set. For more foundational practice, you might find our introductory hybridization guide helpful before tackling the advanced problems below.
2. Solved Examples
Review these detailed solutions to understand the logic behind determining hybridization in complex or non-standard chemical species.
-
Determine the hybridization of the central sulfur atom in Sulfur Hexafluoride (SF6).
-
Count the valence electrons: Sulfur has 6, and each Fluorine has 7. Total = 6 + (6 × 7) = 48 electrons.
-
Draw the structure: Sulfur is the central atom with six single bonds to Fluorine atoms.
-
Calculate the steric number: Sulfur has 6 sigma bonds and 0 lone pairs. Steric Number = 6.
-
Assign hybridization: A steric number of 6 requires six hybrid orbitals. This corresponds to sp3d2 hybridization (1s + 3p + 2d).
-
-
Analyze the hybridization of the oxygen atom in the hydronium ion (H3O+).
-
Count valence electrons: Oxygen (6) + 3 Hydrogen (3) - 1 (positive charge) = 8 electrons.
-
Draw the Lewis structure: Oxygen forms three sigma bonds with Hydrogen and retains one lone pair.
-
Calculate the steric number: 3 bonds + 1 lone pair = 4.
-
Assign hybridization: A steric number of 4 indicates sp3 hybridization.
-
-
Determine the hybridization of the central Iodine in the Triiodide ion (I3-).
-
Count valence electrons: 3 × 7 (Iodine) + 1 (negative charge) = 22 electrons.
-
Draw the structure: The central Iodine is bonded to two terminal Iodine atoms. Remaining electrons (22 - 4 = 18) are distributed as lone pairs.
-
Check the central atom: Central Iodine has 2 sigma bonds and 3 lone pairs.
-
Calculate steric number: 2 + 3 = 5.
-
Assign hybridization: A steric number of 5 corresponds to sp3d hybridization, resulting in a linear molecular geometry despite the trigonal bipyramidal electron arrangement.
-
3. Practice Questions
Test your knowledge with these challenging hybridization problems. Ensure you consider formal charges and resonance where applicable.
1. Identify the hybridization of the central Carbon atom in the Allene molecule (CH2=C=CH2).
2. What is the hybridization of the Xenon atom in Xenon Tetrafluoride (XeF4)?
3. Determine the hybridization of the Nitrogen atom in the Nitronium ion (NO2+).
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. In the molecule ClF3, what is the hybridization of the Chlorine atom?
5. Analyze the Benzyne intermediate (C6H4). What is the hybridization of the two carbons involved in the formal triple bond?
6. Determine the hybridization of the Phosphorus atom in the PCl5 molecule in the gas phase.
7. For the acetate ion (CH3COO-), identify the hybridization of both carbon atoms.
8. What is the hybridization of the central Bromine atom in the BrF5 molecule?
9. Identify the hybridization of the Boron atom in the BF4- anion.
10. Carbon suboxide (C3O2) has a linear structure O=C=C=C=O. Determine the hybridization of all three carbon atoms.
4. Answers & Explanations
Below are the detailed solutions for the practice questions provided above.
-
1. sp: In Allene, the central carbon forms two sigma bonds (one to each adjacent carbon) and two pi bonds. With a steric number of 2, it is sp hybridized.
-
2. sp3d2: Xenon in XeF4 has 4 bonding pairs and 2 lone pairs. Steric number = 6. This requires sp3d2 hybridization to accommodate the octahedral electron geometry.
-
3. sp: The Nitronium ion has a central Nitrogen double-bonded to two Oxygen atoms with no lone pairs. Steric number = 2.
-
4. sp3d: Chlorine in ClF3 has 3 sigma bonds and 2 lone pairs. Steric number = 5. This leads to a T-shaped molecular geometry.
-
5. sp2: In Benzyne, the carbons are part of a ring. While there is a formal triple bond, the geometry is constrained. The "extra" pi bond is formed by the overlap of sp2 orbitals in the plane of the ring, not by unhybridized p-orbitals as in acetylene.
-
6. sp3d: Phosphorus has 5 valence electrons and forms 5 sigma bonds with Chlorine. Steric number = 5.
-
7. sp3 and sp2: The methyl carbon (CH3) has 4 sigma bonds (sp3). The carboxylate carbon has 3 sigma bonds (one to C, two to O) and no lone pairs (sp2).
-
8. sp3d2: Bromine has 7 valence electrons. It forms 5 bonds and has 1 lone pair remaining. Steric number = 6.
-
9. sp3: Boron normally has 3 valence electrons, but the negative charge adds one. With 4 bonds to Fluorine and no lone pairs, the steric number is 4.
-
10. sp: All three carbons in C3O2 are sp hybridized. Each carbon forms two sigma bonds and two pi bonds in a linear arrangement.
5. Quick Quiz
1. Which hybridization state is associated with a trigonal bipyramidal electron-group geometry?
- A sp3
- B sp3d
- C sp3d2
- D sp2
Check answer
Answer: B. sp3d
2. In the molecule Ethyne (C2H2), what is the hybridization of the carbon atoms?
- A sp
- B sp2
- C sp3
- D dsp2
Check answer
Answer: A. sp
3. How many unhybridized p-orbitals remain on an atom with sp2 hybridization?
- A 0
- B 1
- C 2
- D 3
Check answer
Answer: B. 1
4. Which of the following species contains a central atom with sp3d2 hybridization?
- A PCl5
- B SF6
- C CH4
- D BF3
Check answer
Answer: B. SF6
5. What is the steric number of an atom that is sp3 hybridized?
- A 2
- B 3
- C 4
- D 5
Check answer
Answer: C. 4
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →6. Frequently Asked Questions
What is the difference between sp2 and sp3 hybridization?
The primary difference lies in the number of p-orbitals mixed with the s-orbital; sp2 mixes two p-orbitals to form three hybrid orbitals for trigonal planar geometry, while sp3 mixes three p-orbitals to form four hybrid orbitals for tetrahedral geometry. This result is determined by the steric number of the central atom.
Can d-orbitals participate in hybridization for period 2 elements?
No, period 2 elements like Carbon and Nitrogen cannot use d-orbitals for hybridization because they do not have accessible d-subshells in their valence shell. Hybridization involving d-orbitals, such as sp3d, is only observed in elements from period 3 and below, according to Wikipedia's overview of orbital hybridisation.
How does hybridization relate to bond strength?
Hybridization affects bond strength because orbitals with more "s-character" (like sp) hold electrons closer to the nucleus than those with more "p-character" (like sp3). Consequently, sp-hybridized bonds are generally shorter and stronger than sp2 or sp3 bonds between the same types of atoms.
Why is the hybridization of Oxygen in water considered sp3?
Oxygen in water is considered sp3 hybridized because it has a steric number of 4, consisting of two sigma bonds to Hydrogen and two lone pairs. This arrangement follows the LibreTexts Chemistry explanation of VSEPR theory to minimize electron repulsion.
Does resonance affect the hybridization of an atom?
Yes, resonance can affect hybridization because an atom must maintain a consistent hybridization state across all resonance contributors, often requiring an unhybridized p-orbital to participate in pi-bonding or delocalization. In such cases, the atom adopts the hybridization that allows for the necessary p-orbital overlap, such as sp2 instead of sp3.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.