Hybridization Practice Questions with Answers
Concept Explanation
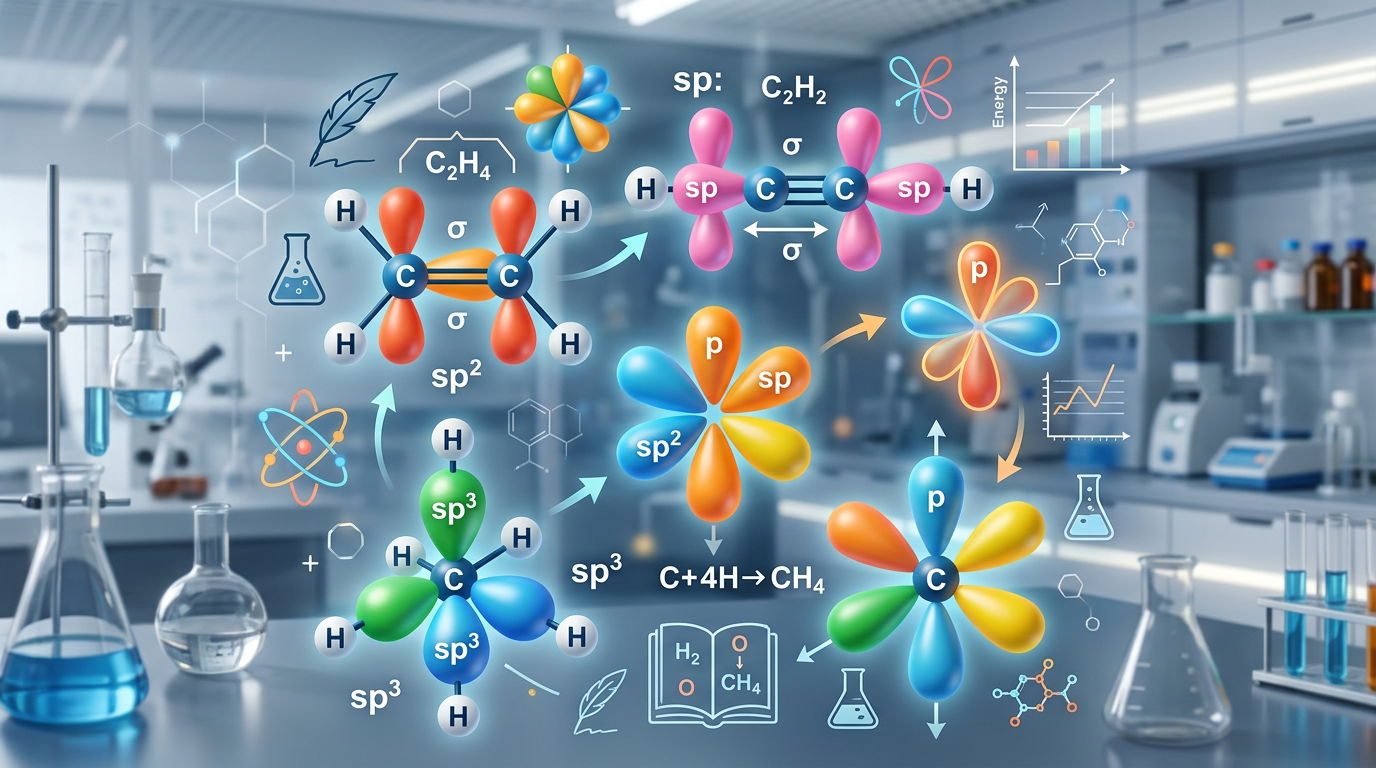
Hybridization is the chemical concept where atomic orbitals within an atom mix to form new, identical hybrid orbitals that facilitate the formation of chemical bonds with specific geometries.
According to Valence Bond Theory, atoms often use a combination of s, p, and sometimes d orbitals to create a set of equivalent orbitals that point in directions that minimize electron repulsion. This process explains why molecules like methane (CH₄) have a tetrahedral shape even though carbon's valence electrons occupy different subshells (2s and 2p). By mixing one 2s orbital and three 2p orbitals, carbon creates four equivalent sp³ hybrid orbitals.
To determine the hybridization of a central atom, we calculate the Steric Number:
Steric Number = (Number of atoms bonded to the central atom) + (Number of lone pairs on the central atom)
Steric Number Hybridization Molecular Geometry Bond Angle 2 sp Linear 180° 3 sp² Trigonal Planar 120° 4 sp³ Tetrahedral 109.5° 5 sp³d Trigonal Bipyramidal 90°, 120° 6 sp³d² Octahedral 90°
Understanding hybridization is essential for predicting molecular reactivity and physical properties. For students mastering broader chemical principles, similar logic is applied when analyzing electronic transitions or calculating reaction rates, much like the steps found in rate law practice questions.
Solved Examples
Example 1: Determine the hybridization of the central atom in Water (H₂O).
-
Identify the central atom: Oxygen (O).
-
Count valence electrons: O has 6, each H has 1. Total = 8.
-
Draw the Lewis structure: Oxygen is bonded to two Hydrogen atoms and has two lone pairs.
-
Calculate Steric Number: 2 bonded atoms + 2 lone pairs = 4.
-
Assign hybridization: A steric number of 4 corresponds to sp³ hybridization.
Example 2: Determine the hybridization of Carbon in Carbon Dioxide (CO₂).
-
Identify the central atom: Carbon (C).
-
Draw the Lewis structure: Carbon forms double bonds with two Oxygen atoms (O=C=O).
-
Calculate Steric Number: Carbon is bonded to 2 atoms and has 0 lone pairs.
-
Assign hybridization: A steric number of 2 corresponds to sp hybridization.
Example 3: Determine the hybridization of Phosphorus in Phosphorus Pentachloride (PCl₅).
-
Identify the central atom: Phosphorus (P).
-
Count valence electrons: P has 5, each Cl has 7.
-
Draw the Lewis structure: P is bonded to 5 Cl atoms with 0 lone pairs on P.
-
Calculate Steric Number: 5 bonded atoms + 0 lone pairs = 5.
-
Assign hybridization: A steric number of 5 corresponds to sp³d hybridization.
Practice Questions
1. What is the hybridization of the Boron atom in Boron Trifluoride (BF₃)?
2. Determine the hybridization of the central Carbon atom in the carbonate ion (CO₃²⁻).
3. Identify the hybridization and the number of lone pairs on the central atom in Ammonia (NH₃).
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. In the molecule Ethene (C₂H₄), what is the hybridization of each Carbon atom?
5. Determine the hybridization of Sulfur in Sulfur Hexafluoride (SF₆).
6. What is the hybridization of Xenon in Xenon Tetrafluoride (XeF₄)?
7. For the molecule Acetylene (C₂H₂), describe the hybridization of the Carbon atoms and the types of bonds (sigma/pi) formed.
8. Calculate the steric number and hybridization for the central Oxygen in the Hydronium ion (H₃O⁺).
9. Identify the hybridization of the central Iodine atom in the Triiodide ion (I₃⁻).
10. Explain why the bond angle in CH₄ is 109.5° while in H₂O it is approximately 104.5°, despite both having the same hybridization.
Answers & Explanations
1. Answer: sp²
Boron has 3 valence electrons and forms 3 single bonds with Fluorine. Steric number = 3 atoms + 0 lone pairs = 3. This corresponds to sp² hybridization and a trigonal planar geometry.
2. Answer: sp²
In CO₃²⁻, the central Carbon is bonded to three Oxygen atoms (one double bond, two single bonds). Steric number = 3 atoms + 0 lone pairs = 3. This results in sp² hybridization.
3. Answer: sp³, 1 lone pair
Nitrogen has 5 valence electrons. It forms 3 bonds with Hydrogen and retains 1 lone pair. Steric number = 3 + 1 = 4. Hybridization is sp³.
4. Answer: sp²
Each Carbon in C₂H₄ is bonded to two Hydrogens and one other Carbon atom (double bond). Steric number = 3 atoms + 0 lone pairs = 3. This is sp² hybridization, allowing for the formation of one pi bond.
5. Answer: sp³d²
Sulfur has 6 valence electrons and bonds with 6 Fluorine atoms. Steric number = 6 atoms + 0 lone pairs = 6. This requires sp³d² hybridization.
6. Answer: sp³d²
Xenon has 8 valence electrons. It bonds with 4 Fluorine atoms and has 2 lone pairs remaining. Steric number = 4 + 2 = 6. Hybridization is sp³d², resulting in a square planar molecular geometry.
7. Answer: sp hybridization; 1 sigma and 2 pi bonds
Each Carbon in C₂H₂ is bonded to one Hydrogen and one Carbon (triple bond). Steric number = 2. The sp hybrid orbitals form sigma bonds, while the two unhybridized p-orbitals form two pi bonds.
8. Answer: Steric Number = 4, sp³
Oxygen in H₃O⁺ is bonded to 3 Hydrogens and has 1 lone pair. Steric number = 3 + 1 = 4. This results in sp³ hybridization.
9. Answer: sp³d
The central Iodine in I₃⁻ is bonded to 2 other Iodine atoms and has 3 lone pairs. Steric number = 2 + 3 = 5. This corresponds to sp³d hybridization.
10. Answer: Lone pair repulsion
Both molecules use sp³ hybrid orbitals. However, H₂O has two lone pairs which exert greater repulsive force than bonding pairs (VSEPR theory), compressing the H-O-H bond angle from the ideal 109.5° to 104.5°.
Quick Quiz
1. Which hybridization corresponds to a linear molecular geometry?
- A sp³
- B sp²
- C sp
- D sp³d
Check answer
Answer: C. sp
2. What is the hybridization of Carbon in Methane (CH₄)?
- A sp
- B sp²
- C sp³
- D sp³d²
Check answer
Answer: C. sp³
3. How many unhybridized p-orbitals remain in an sp² hybridized atom?
- A 0
- B 1
- C 2
- D 3
Check answer
Answer: B. 1
4. A molecule with a steric number of 5 uses which set of hybrid orbitals?
- A sp³
- B sp³d
- C sp³d²
- D sp²
Check answer
Answer: B. sp³d
5. Which molecule contains an sp² hybridized central atom?
- A BeCl₂
- B CH₄
- C BF₃
- D H₂O
Check answer
Answer: C. BF₃
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the difference between sp, sp², and sp³ hybridization?
The difference lies in the number of p-orbitals mixed with the s-orbital: sp mixes one p, sp² mixes two, and sp³ mixes three. This results in different geometries (linear, trigonal planar, and tetrahedral) and different numbers of remaining p-orbitals for pi bonding.
How do you determine hybridization from a Lewis structure?
You determine hybridization by calculating the steric number of the central atom, which is the sum of bonded atoms and lone pairs. This number maps directly to a specific hybrid orbital set, such as 2 for sp or 4 for sp³.
Can lone pairs affect the hybridization of an atom?
Yes, lone pairs occupy hybrid orbitals and must be included in the steric number calculation. While they don't change the hybridization type itself once the steric number is set, they do influence the final molecular geometry and bond angles.
Why does Phosphorus often use sp³d hybridization?
Phosphorus is in the third period of the periodic table, meaning it has access to 3d orbitals. This allows it to expand its octet and form five bonds, requiring the mixing of one s, three p, and one d orbital.
Is hybridization a physical reality or a mathematical model?
Hybridization is a mathematical model within valence bond theory used to explain observed molecular shapes and bond equivalencies. While it provides a useful framework, more complex electronic behaviors are often described using Molecular Orbital Theory.
For more practice with advanced chemical calculations, check out our ideal gas law practice questions or explore reaction order practice questions to further your understanding of chemical kinetics.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.