Easy Strong Acid vs Weak Acid Practice Questions
Concept Explanation
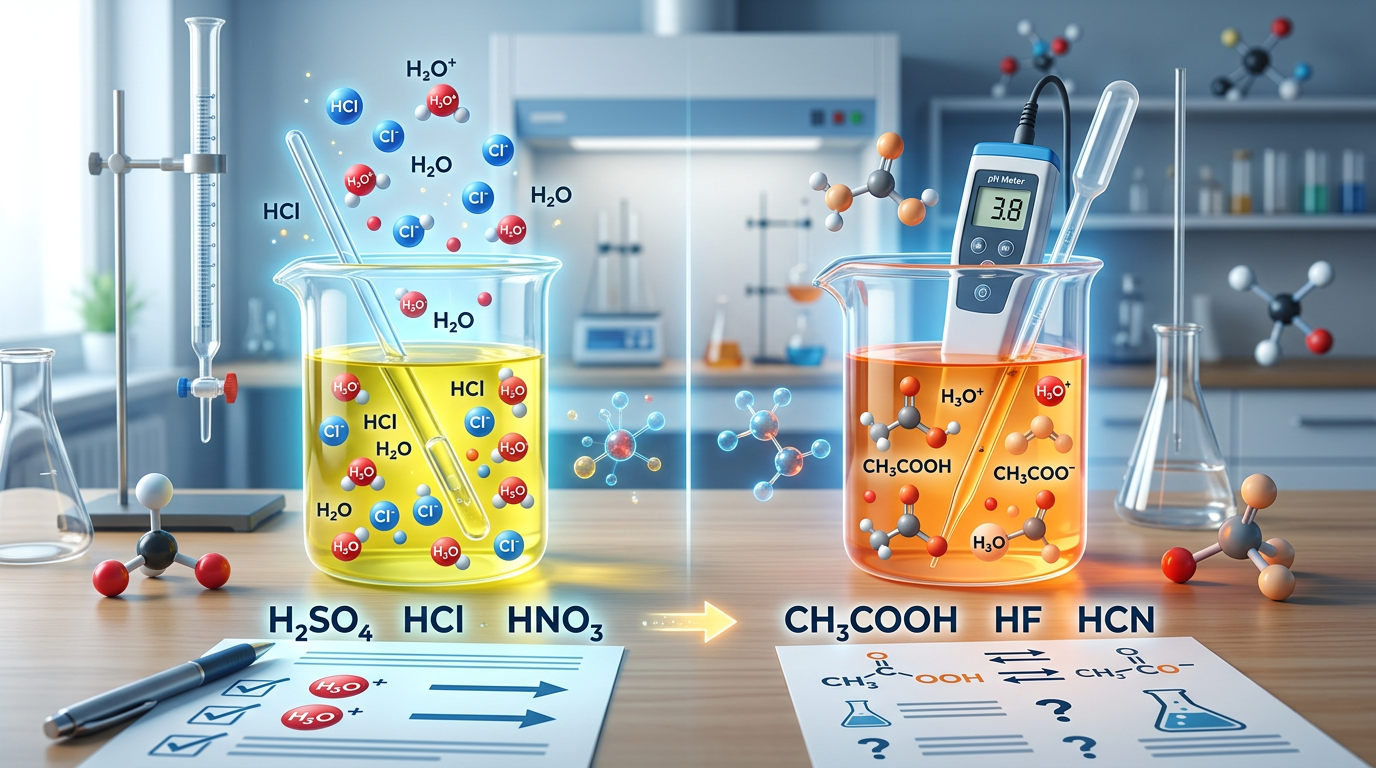
A strong acid is a substance that completely dissociates into its ions in aqueous solution, while a weak acid only partially dissociates, leaving a significant amount of the original molecule intact. This fundamental difference in ionization behavior determines the acid's chemical properties and how it interacts in various reactions. Strong acids, such as hydrochloric acid (HCl), release all their hydrogen ions into the water, resulting in a high concentration of H+ ions. In contrast, weak acids like acetic acid (CH3COOH) exist in an equilibrium state where only a small fraction of the molecules release their protons.
To understand the strength of an acid, scientists often look at the acid dissociation constant (Ka). A high Ka value indicates a strong acid, whereas a low Ka value signifies a weak acid. You can explore more about these values in our Ka and Kb Calculations Practice Questions guide. Another way to distinguish them is through the pH scale; for the same molarity, a strong acid will produce a lower pH than a weak acid because it contributes more hydrogen ions to the solution. This is a critical concept when performing pH Calculation Practice Questions in laboratory settings.
Feature Strong Acid Weak Acid Degree of Ionization ~100% (Complete) < 5% (Partial) Reaction Arrow Single arrow (→) Equilibrium arrows (⇌) Examples HCl, HNO3, H2SO4 HF, CH3COOH, H3PO4
According to the LibreTexts Chemistry resource, there are only seven common strong acids. Memorizing these seven (HCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4) is the easiest way to identify whether an acid is strong or weak; if it is not on this short list, it is almost certainly a weak acid.
Solved Examples
-
Identify if Hydrobromic Acid (HBr) is a strong or weak acid and write its dissociation equation.
-
Check the list of common strong acids. HBr is one of the seven strong mineral acids.
-
Because it is strong, it dissociates completely.
-
Write the equation using a single arrow: HBr(aq) → H+(aq) + Br-(aq).
-
-
Calculate the [H+] concentration in a 0.05 M solution of Nitric Acid (HNO3).
-
Identify HNO3 as a strong acid.
-
Since it dissociates 100%, the concentration of H+ is equal to the initial concentration of the acid.
-
[H+] = 0.05 M.
-
-
Explain why Hydrofluoric Acid (HF) is considered a weak acid even though Fluorine is highly electronegative.
-
Observe the bond strength between Hydrogen and Fluorine. The H-F bond is exceptionally strong.
-
In water, the energy required to break the H-F bond is higher than the energy released by hydration.
-
Consequently, most HF molecules remain intact (HF ⇌ H+ + F-), making it a weak acid.
-
Practice Questions
1. Which of the following acids dissociates completely in water: HCl, HF, or HCN?
2. True or False: A weak acid always has a higher pH than a strong acid, regardless of concentration.
3. Write the chemical equilibrium equation for the dissociation of acetic acid (CH3COOH) in water.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. If you have a 0.1 M solution of HCl and a 0.1 M solution of HF, which solution will have a higher concentration of H+ ions?
5. List three examples of strong acids commonly found in a chemistry laboratory.
6. Why is a double arrow (⇌) used in the dissociation equation of a weak acid?
7. Classify H2CO3 (carbonic acid) as either a strong or weak acid.
8. A solution has a very low Ka value (e.g., 1.8 x 10^-5). Does this represent a strong acid or a weak acid?
9. In a solution of a strong acid, what is the predominant species present: the intact acid molecule or the dissociated ions?
10. How does the electrical conductivity of a 1.0 M solution of HCl compare to a 1.0 M solution of CH3COOH?
Answers & Explanations
-
HCl. Hydrochloric acid is one of the seven strong acids that ionize 100% in water. HF and HCN are weak acids that only partially ionize.
-
False. pH depends on both the strength of the acid and its concentration. A very concentrated weak acid can have a lower pH than an extremely dilute strong acid. For more on this, check out our pH Calculation Practice Questions.
-
CH3COOH ⇌ H+ + CH3COO-. The double arrow signifies that the reaction is reversible and reaches a state of equilibrium where both reactants and products are present.
-
The 0.1 M HCl solution. Because HCl is a strong acid, it produces 0.1 M of H+ ions. HF is a weak acid and will produce significantly less than 0.1 M of H+ ions.
-
HCl, H2SO4, and HNO3. These are standard strong acids used in various chemical syntheses and titrations.
-
To show equilibrium. The double arrow indicates that the acid does not fully break apart; the forward and reverse reactions occur simultaneously, maintaining a balance between the ions and the intact molecules.
-
Weak acid. Carbonic acid is not on the list of common strong acids and only partially dissociates in water, which is vital for the blood buffering system explained in our Buffer Solution Practice Questions.
-
Weak acid. A small Ka value indicates that the equilibrium position lies far to the left, meaning very little product (ions) is formed.
-
The dissociated ions. In a strong acid solution, the original acid molecule (HA) is virtually non-existent because it has all converted into H+ and A-.
-
HCl has higher conductivity. Conductivity depends on the concentration of ions. Since HCl dissociates completely, it provides more ions to carry an electric current than the partially dissociated CH3COOH.
Quick Quiz
1. Which of the following is a characteristic of a strong acid?
- A It has a very small Ka value
- B It dissociates completely in aqueous solution
- C It always has a pH above 7
- D It is represented by a double equilibrium arrow
Check answer
Answer: B. It dissociates completely in aqueous solution
2. Which acid is commonly found in vinegar and serves as a classic example of a weak acid?
- A Sulfuric acid
- B Hydrochloric acid
- C Nitric acid
- D Acetic acid
Check answer
Answer: D. Acetic acid
3. What does a single forward arrow (→) in an acid dissociation equation represent?
- A The reaction is at equilibrium
- B The acid is weak
- C The acid is strong and ionizes completely
- D No ions are formed
Check answer
Answer: C. The acid is strong and ionizes completely
4. If two acid solutions have the same molarity, the one with the higher H+ concentration is:
- A The stronger acid
- B The weaker acid
- C The more dilute acid
- D The one with the higher pKa
Check answer
Answer: A. The stronger acid
5. Which of these is NOT one of the seven common strong acids?
- A HI
- B HClO4
- C H3PO4
- D HNO3
Check answer
Answer: C. H3PO4
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the main difference between a strong acid and a weak acid?
The main difference lies in the degree of ionization; strong acids dissociate 100% into ions in water, whereas weak acids only partially dissociate, existing in equilibrium with their molecular form.
Does the concentration of an acid determine if it is strong or weak?
No, strength is an intrinsic property of the acid's molecular structure and its tendency to lose a proton, while concentration refers to the amount of acid dissolved in a specific volume of solvent.
Can a weak acid be more dangerous than a strong acid?
Yes, some weak acids like Hydrofluoric acid (HF) are extremely dangerous because they can penetrate skin and react with calcium in bones, regardless of their partial dissociation in water.
How can I tell if an acid is strong just by looking at its formula?
The most reliable method is to memorize the seven common strong acids (HCl, HBr, HI, HNO3, H2SO4, HClO3, HClO4); if an acid is not on this list, it is generally classified as weak.
Why do we use Ka values for weak acids but not usually for strong acids?
Weak acids reach an equilibrium that can be measured by a constant, whereas strong acids dissociate so completely that the Ka value is effectively infinite, making it less useful for standard calculations.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.