Easy Redox Reaction Practice Questions
Concept Explanation
A redox reaction, short for reduction-oxidation reaction, is a chemical process characterized by the transfer of electrons between two species. In these reactions, one substance loses electrons (oxidation) while another substance gains those same electrons (reduction). You can easily remember this using the mnemonic "LEO the lion says GER": Loss of Electrons is Oxidation, and Gain of Electrons is Reduction.
To identify these changes, chemists assign oxidation numbers to atoms within molecules or ions. An oxidation number is a theoretical charge an atom would have if all bonds were 100% ionic. When the oxidation number of an element increases during a reaction, it has been oxidized. Conversely, if the oxidation number decreases, it has been reduced. For those looking to master the basics, starting with Redox Reaction Practice Questions with Answers is an excellent way to build a foundation.
Redox reactions are the driving force behind many essential processes, from the metabolism of food in our bodies to the generation of electricity in batteries. According to Wikipedia, these reactions are central to both biological systems and industrial applications. Understanding how to track electron flow is the first step toward calculating complex values like cell potentials or using the Nernst Equation.
Key Terms to Know:
-
Oxidizing Agent: The substance that gains electrons and becomes reduced. It "oxidizes" the other reactant.
-
Reducing Agent: The substance that loses electrons and becomes oxidized. It "reduces" the other reactant.
-
Half-Reaction: An equation that shows either the oxidation or the reduction component of a redox reaction separately.
Solved Examples
Reviewing these step-by-step examples will help you understand how to approach easy redox reaction practice questions with confidence.
Example 1: Simple Metal Displacement
Reaction: Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
-
Assign oxidation numbers: Mg is 0 (elemental), H in HCl is +1, Cl is -1. In MgCl₂, Mg is +2 and Cl is -1. H₂ is 0.
-
Identify changes: Mg goes from 0 to +2 (increase). H goes from +1 to 0 (decrease).
-
Determine roles: Mg is oxidized (reducing agent). H⁺ is reduced (oxidizing agent).
Example 2: Formation of an Ionic Compound
Reaction: 2Na(s) + Cl₂(g) → 2NaCl(s)
-
Assign oxidation numbers: Na and Cl₂ are both 0 as pure elements. In NaCl, Na is +1 and Cl is -1.
-
Identify changes: Na increases from 0 to +1. Cl decreases from 0 to -1.
-
Determine roles: Na is the reducing agent; Cl₂ is the oxidizing agent.
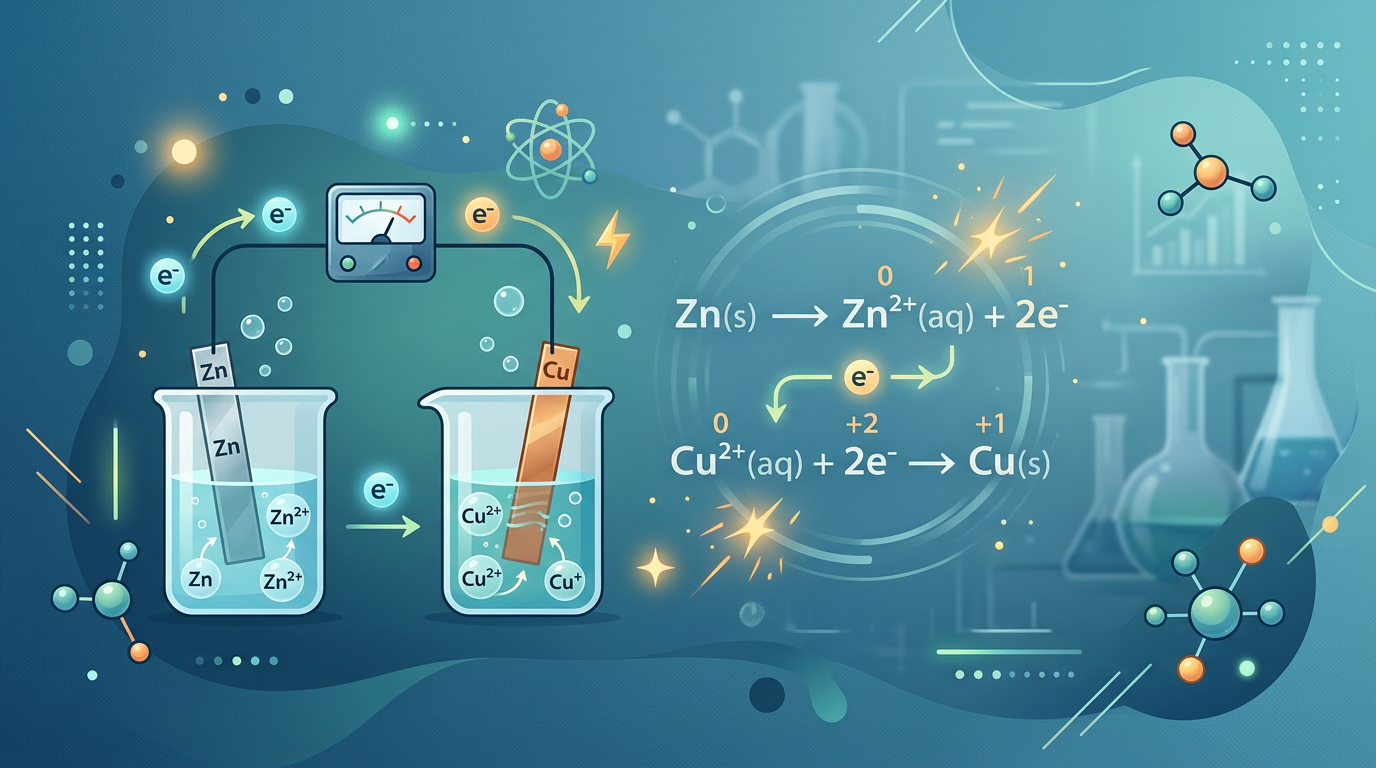
Example 3: Identifying Half-Reactions
Reaction: Zn + Cu²⁺ → Zn²⁺ + Cu
-
Split the reaction into two parts based on the elements involved.
-
Oxidation half-reaction: Zn → Zn²⁺ + 2e⁻ (Loss of electrons).
-
Reduction half-reaction: Cu²⁺ + 2e⁻ → Cu (Gain of electrons).
3. **Practice Questions**
Test your knowledge with these easy redox reaction practice questions. The difficulty increases slightly as you progress.
1. In the reaction 2Li + F₂ → 2LiF, which element is being oxidized?
2. Identify the oxidizing agent in the following reaction: Fe + CuSO₄ → FeSO₄ + Cu.
3. What is the oxidation number of Oxygen in its elemental form (O₂)?
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. In the reaction 4Al + 3O₂ → 2Al₂O₃, which species acts as the reducing agent?
5. Assign oxidation numbers to all atoms in the molecule H₂SO₄.
6. True or False: In a redox reaction, the number of electrons lost must equal the number of electrons gained.
7. Identify the reduction half-reaction for: 2Ag⁺ + Cu → 2Ag + Cu²⁺.
8. What is the oxidation state of Manganese in KMnO₄?
9. Which of the following is NOT a redox reaction?
a) NaOH + HCl → NaCl + H₂O
b) 2H₂ + O₂ → 2H₂O
10. If an atom's oxidation number changes from +3 to +5, has it been oxidized or reduced?
Answers & Explanations
-
Lithium (Li). Lithium starts at 0 and goes to +1 in LiF. Since its oxidation number increased, it lost electrons and was oxidized.
-
Cu²⁺ (from CuSO₄). Copper goes from +2 in CuSO₄ to 0 in its solid form. Since it gained electrons, it is reduced, making it the oxidizing agent.
-
0. Atoms in their pure elemental form always have an oxidation number of zero, as per LibreTexts Chemistry standards.
-
Aluminum (Al). Aluminum's oxidation state increases from 0 to +3. By losing electrons, it reduces the oxygen, making Al the reducing agent.
-
H = +1, S = +6, O = -2. Each H is +1 (total +2), each O is -2 (total -8). To make the neutral molecule sum to zero, S must be +6.
-
True. Charge must be conserved in every chemical reaction; electrons are transferred, not destroyed.
-
2Ag⁺ + 2e⁻ → 2Ag. Silver ions gain electrons to become neutral silver atoms.
-
+7. K is +1, each O is -2 (total -8). 1 + x + (-8) = 0, so x = +7.
-
a) NaOH + HCl → NaCl + H₂O. This is an acid-base neutralization. The oxidation states of Na (+1), O (-2), H (+1), and Cl (-1) remain unchanged throughout.
-
Oxidized. An increase in oxidation number indicates a loss of electrons.
Quick Quiz
1. Which mnemonic is commonly used to remember the definition of oxidation and reduction?
- A PV = nRT
- B LEO says GER
- C ROY G BIV
- D SOH CAH TOA
Check answer
Answer: B. LEO says GER
2. In a redox reaction, what happens to the reducing agent?
- A It gains electrons
- B It is oxidized
- C Its oxidation number decreases
- D It remains unchanged
Check answer
Answer: B. It is oxidized
3. What is the oxidation number of any uncombined element?
- A +1
- B -1
- C 0
- D +2
- E -2
Check answer
Answer: C. 0
4. Which of these reactions involves the transfer of electrons?
- A Precipitation
- B Redox
- C Acid-Base Neutralization
- D Double Displacement
Check answer
Answer: B. Redox
5. If a substance is a strong oxidizing agent, it has a high tendency to:
- A Lose protons
- B Lose electrons
- C Gain electrons
- D Become a noble gas
Check answer
Answer: C. Gain electrons
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the easiest way to identify a redox reaction?
The easiest way to identify a redox reaction is to look for changes in oxidation numbers of the elements from reactants to products. If any element's oxidation state increases or decreases, a redox process has occurred.
Can a reaction be oxidation without reduction?
No, oxidation and reduction must always occur simultaneously because electrons lost by one species must be gained by another. This conservation of charge is a fundamental principle of chemistry.
How do I find the oxidation number of an element in a compound?
Assign known values first, such as +1 for Group 1 metals and -2 for Oxygen, then ensure the sum of all oxidation numbers equals the total charge of the molecule or ion. This algebraic approach allows you to solve for the unknown element.
What is a disproportionation reaction?
A disproportionation reaction is a specific type of redox reaction where the same element is both oxidized and reduced. A common example is the decomposition of hydrogen peroxide into water and oxygen gas.
Why are redox reactions important in everyday life?
Redox reactions power the world by enabling energy transfer in biological respiration and providing the electrical current in devices like smartphones and electric vehicles. They are also responsible for natural processes like the rusting of iron and photosynthesis.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.