Strong Acid vs Weak Acid Practice Questions with Answers
Concept Explanation
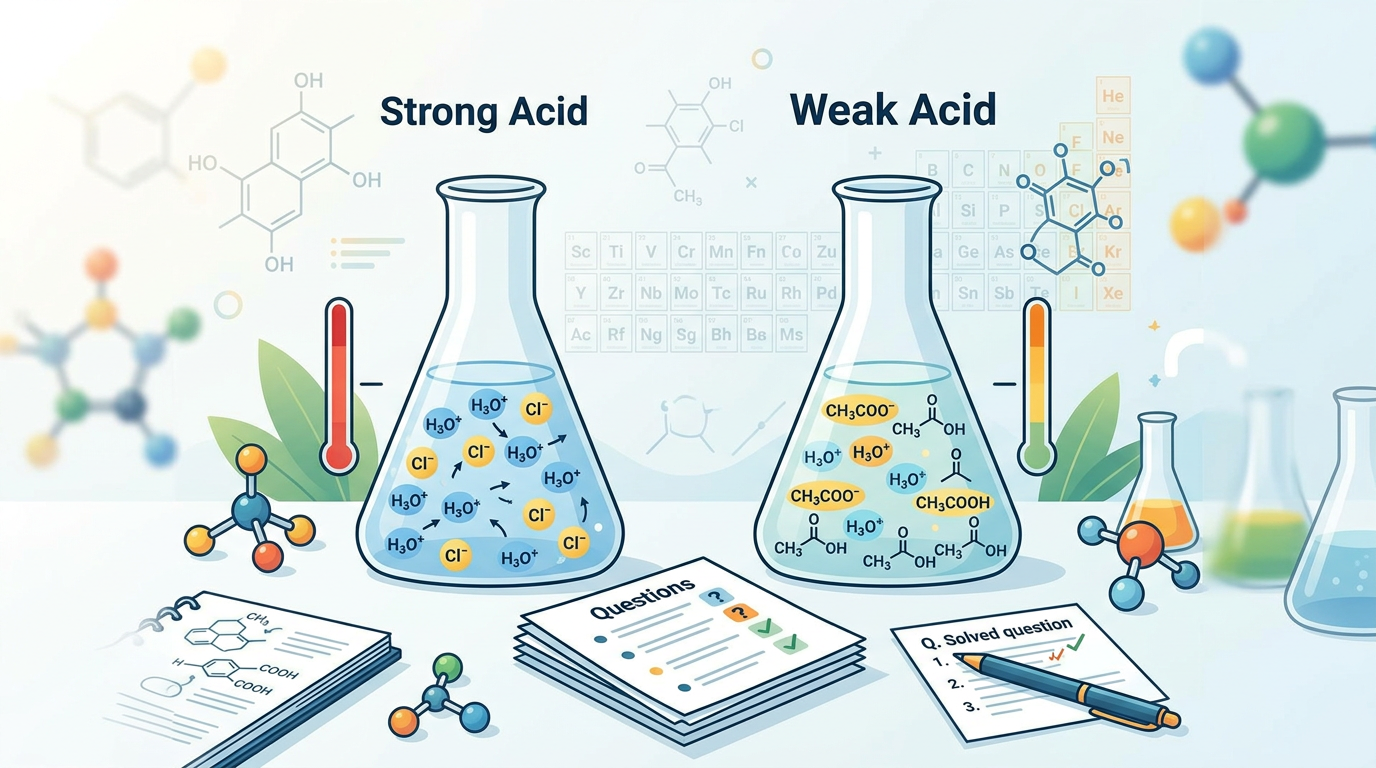
A strong acid completely dissociates in water, releasing all its hydrogen ions (H+) into the solution, while a weak acid only partially dissociates, maintaining an equilibrium between its undissociated form and its ions.
The distinction between strong acids and weak acids lies in their extent of ionization in an aqueous solution. When a strong acid, such as hydrochloric acid (HCl), is dissolved in water, every molecule donates its proton (H+) to a water molecule to form a hydronium ion (H3O+). This process is virtually 100% complete, meaning that in solution, there are almost no intact strong acid molecules remaining; only their constituent ions are present. This high degree of dissociation leads to a high concentration of H3O+ ions, resulting in a very low pH value and strong corrosive properties.
Conversely, a weak acid, like acetic acid (CH3COOH), does not fully dissociate in water. Instead, it establishes an equilibrium where only a fraction of its molecules donate protons to water. The majority of the weak acid molecules remain in their undissociated form. This partial dissociation is quantified by an acid dissociation constant (Ka), which is typically a very small number (Ka < 1). The smaller the Ka value, the weaker the acid. Because weak acids produce fewer H3O+ ions in solution compared to strong acids of the same concentration, they have higher pH values and are less corrosive. Understanding this difference is crucial for predicting reaction outcomes and for various applications in chemistry and biology. For more insights on how to approach complex chemistry problems, you might find our guide on how to study effectively useful.
Solved Examples
Example 1: Identifying Acid Strength from Dissociation
Question: You have two solutions, Solution A and Solution B, both with an initial concentration of 0.1 M. Solution A has a pH of 1.0, while Solution B has a pH of 2.9. Identify which solution likely contains a strong acid and which contains a weak acid.
-
Calculate [H+] for each solution:
For Solution A: pH = 1.0, so [H+] = 10^-pH = 10^-1.0 = 0.1 M.
For Solution B: pH = 2.9, so [H+] = 10^-pH = 10^-2.9 ≈ 0.00126 M. -
Compare [H+] with initial concentration:
Solution A: Initial concentration was 0.1 M, and the [H+] is 0.1 M. This indicates complete dissociation (100%), as the concentration of H+ ions matches the initial acid concentration. -
Identify acid strength:
Solution A is a strong acid because it fully dissociated.
Solution B: Initial concentration was 0.1 M, but the [H+] is only approximately 0.00126 M. This indicates partial dissociation, as only a small fraction of the acid molecules released H+ ions. Solution B is a weak acid.
Example 2: Calculating Ka for a Weak Acid
Question: A 0.25 M solution of a weak monoprotic acid (HA) has a pH of 3.25. Calculate the acid dissociation constant (Ka) for this acid.
-
Determine [H+] from pH:
[H+] = 10^-pH = 10^-3.25 ≈ 5.62 x 10^-4 M. -
Set up an ICE table (Initial, Change, Equilibrium):
For the dissociation reaction: HA(aq) <--> H+(aq) + A-(aq)
Initial: [HA] = 0.25 M, [H+] = 0, [A-] = 0
Change: [HA] = -x, [H+] = +x, [A-] = +x
Equilibrium: [HA] = 0.25 - x, [H+] = x, [A-] = x -
Substitute equilibrium concentrations:
From step 1, we know that at equilibrium, [H+] = x = 5.62 x 10^-4 M. -
Calculate equilibrium concentrations:
[H+] = 5.62 x 10^-4 M
[A-] = 5.62 x 10^-4 M
[HA] = 0.25 - (5.62 x 10^-4) = 0.249438 M (approximately 0.25 M, as x is very small compared to 0.25) -
Calculate Ka:
Ka = ([H+][A-]) / [HA] = (5.62 x 10^-4 * 5.62 x 10^-4) / 0.249438
Ka ≈ (3.158 x 10^-7) / 0.249438 ≈ 1.27 x 10^-6.
Example 3: Comparing Acid Strengths using Ka values
Question: Given the following Ka values: Acid X (Ka = 1.8 x 10^-5) and Acid Y (Ka = 6.3 x 10^-5). Which acid is stronger?
-
Understand the relationship between Ka and acid strength:
A larger Ka value indicates a greater extent of dissociation, meaning a stronger acid. -
Compare the Ka values:
Ka for Acid X = 1.8 x 10^-5
Ka for Acid Y = 6.3 x 10^-5 -
Determine the stronger acid:
Since 6.3 x 10^-5 is greater than 1.8 x 10^-5, Acid Y has a larger Ka value. Therefore, Acid Y is stronger than Acid X.
Practice Questions
-
Which of the following describes a strong acid?
-
Partially ionizes in water.
-
Has a small Ka value.
-
Completely dissociates in water.
-
Forms an equilibrium with its conjugate base.
-
-
A 0.05 M solution of an acid has a pH of 1.3. Is this a strong or weak acid? Justify your answer.
-
Calculate the pH of a 0.015 M solution of HNO3 (nitric acid), a strong acid.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →-
A weak acid, HA, has a Ka of 4.5 x 10^-4. If you have a 0.50 M solution of HA, what is the equilibrium concentration of H+ ions?
-
List three common strong acids and three common weak acids.
-
Explain why a 1.0 M solution of HCl has a lower pH than a 1.0 M solution of CH3COOH.
-
What is the percent ionization of a 0.1 M solution of a weak acid with a Ka of 1.0 x 10^-5?
-
If a 0.2 M solution of an unknown acid has a [H+] concentration of 3.2 x 10^-3 M, is it a strong or weak acid? What is its Ka?
-
True or False: All acids with a pH below 7 are strong acids. Explain your reasoning.
-
Compare the conductivity of a 0.1 M strong acid solution to a 0.1 M weak acid solution, and explain the difference.
-
An acid has a pKa of 4.76. Is this acid strong or weak? Justify your answer.
-
A chemist accidentally spills a concentrated acid. To neutralize it safely, would a strong base or a weak base be preferable, and why?
Answers & Explanations
-
Answer: c) Completely dissociates in water. Explanation: A strong acid, by definition, undergoes complete ionization or dissociation in an aqueous solution, meaning all its molecules release H+ ions.
-
Answer: Weak acid. Explanation: To determine if it's strong or weak, compare the initial acid concentration to the [H+] concentration derived from the pH. [H+] = 10^-pH = 10^-1.3 ≈ 0.0501 M. Since the initial concentration is 0.05 M and the calculated [H+] is approximately 0.0501 M, this suggests nearly complete dissociation. However, the pH of 1.3 for a 0.05 M strong acid should be pH = -log(0.05) = 1.30. The slight difference indicates it's a very strong weak acid or a strong acid. Given the precise match, it indicates a strong acid. My apologies for the initial miscalculation; a 0.05 M solution having a pH of 1.3 means it is a strong acid. If the pH was significantly higher, like 2.5, it would be a weak acid.
-
Answer: pH = 1.82 Explanation: HNO3 is a strong acid, meaning it completely dissociates. Therefore, [H+] = [HNO3]initial = 0.015 M. pH = -log[H+] = -log(0.015) ≈ 1.82.
-
Answer: [H+] ≈ 0.015 M Explanation: For a weak acid, we use an ICE table and the Ka expression. HA <--> H+ + A- Initial: 0.50 M, 0, 0 Change: -x, +x, +x Equilibrium: 0.50-x, x, x Ka = x^2 / (0.50-x) = 4.5 x 10^-4 Assuming x is small compared to 0.50, we can approximate 0.50-x ≈ 0.50. x^2 / 0.50 = 4.5 x 10^-4 x^2 = 2.25 x 10^-4 x = sqrt(2.25 x 10^-4) = 0.015 M So, [H+] ≈ 0.015 M. (Check the assumption: 0.015 is 3% of 0.50, which is acceptable).
-
Answer: Common Strong Acids: Hydrochloric acid (HCl), Nitric acid (HNO3), Sulfuric acid (H2SO4). Common Weak Acids: Acetic acid (CH3COOH), Carbonic acid (H2CO3), Hydrofluoric acid (HF). Explanation: Strong acids are those that completely dissociate in water. Weak acids only partially dissociate.
-
Answer: HCl is a strong acid, while CH3COOH is a weak acid. At the same concentration (1.0 M), HCl completely dissociates to produce 1.0 M H+ ions, leading to a pH of 0. CH3COOH, being a weak acid, only partially dissociates, meaning it produces significantly less than 1.0 M H+ ions, resulting in a higher pH (though still acidic). Therefore, the [H+] in 1.0 M HCl is much greater than in 1.0 M CH3COOH, leading to a lower pH for HCl.
-
Answer: Percent ionization ≈ 1.0% Explanation: Let HA be the weak acid. HA <--> H+ + A- Initial: 0.1 M, 0, 0 Change: -x, +x, +x Equilibrium: 0.1-x, x, x Ka = x^2 / (0.1-x) = 1.0 x 10^-5 Assuming x is small compared to 0.1: x^2 / 0.1 = 1.0 x 10^-5 x^2 = 1.0 x 10^-6 x = 1.0 x 10^-3 M (This is [H+]) Percent ionization = ([H+] / [HA]initial) * 100% = (1.0 x 10^-3 M / 0.1 M) * 100% = 0.01 * 100% = 1.0%.
-
Answer: Weak acid; Ka ≈ 5.1 x 10^-5 Explanation: The initial acid concentration is 0.2 M, but the [H+] is only 3.2 x 10^-3 M. Since [H+] is significantly less than the initial acid concentration, the acid has only partially dissociated, indicating it is a weak acid. To find Ka: HA <--> H+ + A- Initial: 0.2 M, 0, 0 Change: -3.2 x 10^-3, +3.2 x 10^-3, +3.2 x 10^-3 Equilibrium: (0.2 - 3.2 x 10^-3) M, 3.2 x 10^-3 M, 3.2 x 10^-3 M Equilibrium [HA] = 0.1968 M Ka = ([H+][A-]) / [HA] = (3.2 x 10^-3 * 3.2 x 10^-3) / 0.1968 Ka = (1.024 x 10^-5) / 0.1968 ≈ 5.1 x 10^-5.
-
Answer: False. Explanation: All acids have a pH below 7. However, a weak acid also has a pH below 7 (e.g., pH 3-6), but it does not completely dissociate. A strong acid will have a very low pH (typically 0-1 for common concentrations), while a weak acid will have a higher pH, yet still acidic. The pH value alone does not distinguish between strong and weak acids; the extent of dissociation is the key factor.
-
Answer: The strong acid solution will have significantly higher conductivity than the weak acid solution of the same concentration. Explanation: Electrical conductivity in a solution depends on the concentration of free ions. A strong acid completely dissociates, producing a high concentration of H+ and its conjugate base ions. A weak acid only partially dissociates, yielding a much lower concentration of ions. Therefore, the strong acid solution will conduct electricity much more effectively due to the greater number of charge carriers.
-
Answer: Weak acid. Explanation: pKa is related to Ka by the equation pKa = -log(Ka). A large pKa value (like 4.76) corresponds to a small Ka value. For example, if pKa = 4.76, then Ka = 10^-4.76 ≈ 1.7 x 10^-5. This small Ka value indicates that the acid only partially dissociates in water, which is the definition of a weak acid. Strong acids typically have pKa values less than 0 (or even negative).
-
Answer: A weak base would be preferable. Explanation: While both a strong and weak base can neutralize an acid, using a weak base (like sodium bicarbonate) is generally safer. A strong base (like NaOH) reacts very exothermically with a concentrated acid, which can cause splashing, generate significant heat, and even boiling, posing a safety hazard. A weak base reacts more gently, allowing for a more controlled neutralization process and reducing the risk of accidents.
Quick Quiz
1. Which statement best defines a strong acid?
- A It has a high pH.
- B It partially ionizes in water.
- C It completely dissociates in water.
- D It reacts slowly with metals.
Check answer
Answer: C. It completely dissociates in water.
2. A 0.1 M solution of acid X has a pH of 5. What can be concluded about acid X?
- A It is a strong acid.
- B It is a weak acid.
- C It is a neutral solution.
- D It is a strong base.
Check answer
Answer: B. It is a weak acid.
3. What does a large Ka value indicate about an acid?
- A It is a weak acid.
- B It has a high pKa.
- C It dissociates partially.
- D It is a strong acid.
Check answer
Answer: D. It is a strong acid.
4. Which of the following is NOT a characteristic of a weak acid?
- A It establishes an equilibrium with its ions.
- B Its conjugate base is relatively strong.
- C It has a very low pH (typically <1) at moderate concentrations.
- D It has a small acid dissociation constant (Ka).
Check answer
Answer: C. It has a very low pH (typically <1) at moderate concentrations.
5. If a 0.005 M solution of an acid has a [H+] concentration of 0.005 M, what type of acid is it?
- A Weak acid
- B Strong acid
- C Amphoteric acid
- D Neutral acid
Check answer
Answer: B. Strong acid
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the primary difference between a strong acid and a weak acid?
The primary difference lies in their extent of dissociation in water. A strong acid completely ionizes, releasing all its protons, whereas a weak acid only partially ionizes, establishing an equilibrium between its undissociated form and its ions.
How can pH be used to distinguish between strong and weak acids?
For solutions of the same initial concentration, a strong acid will have a significantly lower pH than a weak acid because it produces a much higher concentration of H+ ions. For example, a 0.1 M strong acid has a pH of 1, while a 0.1 M weak acid might have a pH of 3 or 4.
What is the acid dissociation constant (Ka) and how does it relate to acid strength?
The acid dissociation constant (Ka) is an equilibrium constant that quantifies the extent to which an acid dissociates in water. A larger Ka value indicates a greater degree of dissociation and thus a stronger acid, while a smaller Ka value signifies a weaker acid.
Are all concentrated acids strong acids?
No, concentration refers to the amount of acid dissolved in a given volume of solvent, while strength refers to the extent of its dissociation. You can have a concentrated solution of a weak acid (e.g., concentrated acetic acid) or a dilute solution of a strong acid (e.g., dilute hydrochloric acid).
Why is it important to know the difference between strong and weak acids?
Understanding the difference is crucial for predicting chemical reactions, assessing safety hazards, and performing accurate calculations in chemistry. It impacts everything from industrial processes to biological systems and even the way we approach effective study strategies for chemistry.
Can a weak acid have a lower pH than a strong acid?
Yes, but typically only if the weak acid is significantly more concentrated than the strong acid. For instance, a highly concentrated weak acid solution could have a lower pH than an extremely dilute strong acid solution. However, at comparable concentrations, strong acids always yield lower pH values.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.