Polarity Determination Practice Questions with Answers
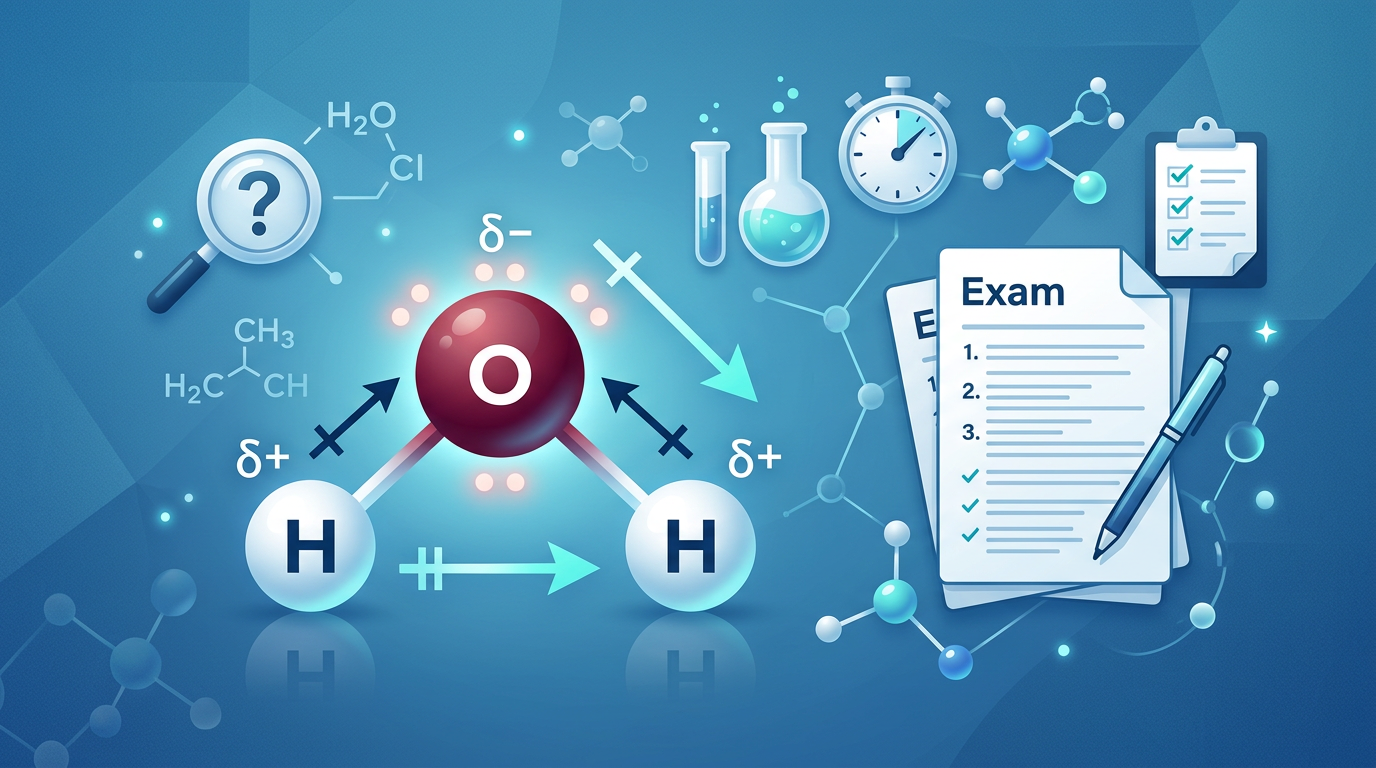
Concept Explanation
Polarity determination is the process of identifying whether a molecule possesses a net dipole moment based on its molecular geometry and the electronegativity differences between its constituent atoms. To determine if a molecule is polar or nonpolar, you must evaluate two primary factors: the presence of polar covalent bonds and the spatial arrangement of those bonds. A polar bond occurs when electrons are shared unequally between atoms with different electronegativity values, typically cited from the Pauling scale. However, the existence of polar bonds does not guarantee a polar molecule; if the bond dipoles are equal in magnitude and point in opposite directions, they cancel out, resulting in a nonpolar molecule (like CO₂). Conversely, if the geometry is asymmetrical (like H₂O), the dipoles do not cancel, creating a net molecular dipole.
The process generally follows these steps:
-
Draw the Lewis Structure: Identify the valence electrons and arrange them to satisfy the octet rule.
-
Determine Molecular Geometry: Use Valence Shell Electron Pair Repulsion (VSEPR) theory to find the 3D shape.
-
Evaluate Bond Polarity: Calculate the electronegativity difference (ΔEN). Generally, ΔEN > 0.4 indicates a polar bond.
-
Sum the Dipole Vectors: Determine if the symmetry of the shape allows the dipoles to cancel.
Understanding polarity is essential for predicting physical properties like boiling points, solubility, and reactivity. For students mastering related chemical concepts, practicing with redox reaction practice questions or exploring ideal gas law practice questions can provide a broader context for how molecular interactions affect macroscopic behavior.
Solved Examples
Review these step-by-step solutions to understand how to apply VSEPR and electronegativity rules to determine molecular polarity.
Example 1: Carbon Dioxide (CO₂)
-
Lewis Structure: Carbon is the central atom with double bonds to each Oxygen (O=C=O).
-
Geometry: The molecule has two electron domains and no lone pairs on the central atom, making it Linear.
-
Bond Polarity: Oxygen (3.44) is more electronegative than Carbon (2.55). The C=O bonds are polar.
-
Net Dipole: Because the molecule is linear and the two dipoles point in exactly opposite directions, they cancel out. Result: Nonpolar.
Example 2: Water (H₂O)
-
Lewis Structure: Oxygen is the central atom with two single bonds to Hydrogen and two lone pairs.
-
Geometry: Four electron domains (2 bonding, 2 lone pairs) result in a Bent shape.
-
Bond Polarity: Oxygen is significantly more electronegative than Hydrogen.
-
Net Dipole: The bent shape means the dipoles do not cancel; they combine to point toward the oxygen atom. Result: Polar.
Example 3: Boron Trifluoride (BF₃)
-
Lewis Structure: Boron is the central atom with three single bonds to Fluorine atoms (an exception to the octet rule).
-
Geometry: Three electron domains result in a Trigonal Planar shape.
-
Bond Polarity: B-F bonds are highly polar due to Fluorine's high electronegativity.
-
Net Dipole: In a perfectly symmetrical trigonal planar arrangement with identical outer atoms, the dipoles cancel. Result: Nonpolar.
Practice Questions
Test your knowledge with these polarity determination practice questions ranging from easy to hard.
1. Determine the polarity of CH₄ (Methane) using its molecular geometry.
2. Is NH₃ (Ammonia) polar or nonpolar? Explain based on its lone pairs.
3. Analyze CCl₄ (Carbon Tetrachloride). Does it have a net dipole moment?
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. Determine the polarity of SO₂ (Sulfur Dioxide).
5. Predict the polarity of CH₂Cl₂ (Dichloromethane).
6. Evaluate PF₅ (Phosphorus Pentafluoride). Is it polar?
7. Consider SF₄ (Sulfur Tetrafluoride). Based on its see-saw shape, is it polar?
8. Determine the polarity of XeF₄ (Xenon Tetrafluoride) considering its square planar geometry.
9. Is HF (Hydrogen Fluoride) polar or nonpolar?
10. Analyze BeCl₂ (Beryllium Chloride) in the gas phase.
Answers & Explanations
1. CH₄: Nonpolar. Methane has a tetrahedral geometry. Although C-H bonds have a slight polarity, the symmetrical arrangement of the four identical bonds causes the dipoles to cancel out completely.
2. NH₃: Polar. Nitrogen has one lone pair and three bonding pairs, creating a trigonal pyramidal shape. The lone pair disrupts the symmetry, ensuring the N-H bond dipoles do not cancel, leading to a net dipole toward the lone pair.
3. CCl₄: Nonpolar. Like methane, carbon tetrachloride is tetrahedral. Even though C-Cl bonds are quite polar, the four bonds are identical and arranged symmetrically, resulting in zero net dipole.
4. SO₂: Polar. Sulfur dioxide has a bent molecular geometry due to the lone pair on the sulfur atom. This asymmetry prevents the S-O bond dipoles from canceling.
5. CH₂Cl₂: Polar. While the shape is tetrahedral, the outer atoms are not identical (2 Hydrogens, 2 Chlorines). The C-Cl dipoles are much stronger than C-H, and they do not cancel out.
6. PF₅: Nonpolar. This molecule adopts a trigonal bipyramidal geometry. The three equatorial bonds cancel each other, and the two axial bonds cancel each other, leaving no net dipole.
7. SF₄: Polar. Sulfur has four bonding pairs and one lone pair, resulting in a see-saw shape. This irregular geometry means the polar S-F bonds do not cancel out.
8. XeF₄: Nonpolar. Xenon has four bonds and two lone pairs. The lone pairs are opposite each other (axial), and the four fluorines are in a plane (square planar). The symmetry allows all dipoles to cancel.
9. HF: Polar. As a diatomic molecule with two different atoms, there is no symmetry to cancel the electronegativity difference. Fluorine pulls electron density toward itself.
10. BeCl₂: Nonpolar. Beryllium forms two single bonds with Chlorine in a linear arrangement. The two identical polar bonds point in opposite directions and cancel.
Quick Quiz
1. Which molecular geometry usually results in a nonpolar molecule if all terminal atoms are identical?
- A Bent
- B Trigonal Pyramidal
- C Tetrahedral
- D See-saw
Check answer
Answer: C. Tetrahedral
2. Why is water (H₂O) a polar molecule?
- A It has a linear shape
- B The bond dipoles cancel each other out
- C It has a bent geometry due to lone pairs on oxygen
- D Hydrogen is more electronegative than oxygen
Check answer
Answer: C. It has a bent geometry due to lone pairs on oxygen
3. Which of the following molecules contains polar bonds but is a nonpolar molecule?
- A NH₃
- B CCl₄
- C CHCl₃
- D H₂O
Check answer
Answer: B. CCl₄
4. In a trigonal bipyramidal molecule like PF₅, why is it nonpolar?
- A The bonds are nonpolar
- B The symmetry of the axial and equatorial positions cancels the dipoles
- C Phosphorus has a lone pair
- D The atoms have the same electronegativity
Check answer
Answer: B. The symmetry of the axial and equatorial positions cancels the dipoles
5. What is the primary tool used to predict the 3D shape of a molecule for polarity determination?
- A Ideal Gas Law
- B Hess's Law
- C VSEPR Theory
- D The Nernst Equation
Check answer
Answer: C. VSEPR Theory
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
Can a molecule with polar bonds be nonpolar?
Yes, if the molecule is highly symmetrical and the surrounding atoms are identical, the individual bond dipoles will cancel each other out. Examples include Carbon Dioxide (CO₂) and Carbon Tetrachloride (CCl₄).
How does electronegativity affect polarity?
Electronegativity determines how strongly an atom attracts shared electrons. A large difference in electronegativity between two atoms creates a polar bond, which is the first requirement for a polar molecule.
Is every asymmetrical molecule polar?
Generally, yes; if a molecule lacks symmetry due to different terminal atoms or the presence of lone pairs on the central atom, the bond dipoles will not cancel, resulting in a polar molecule.
What is the difference between bond polarity and molecular polarity?
Bond polarity refers to the electron distribution between two specific atoms, while molecular polarity is the vector sum of all bond dipoles in the entire 3D structure. You can learn more about related chemical properties in our balancing redox practice questions.
Are lone pairs always a sign of a polar molecule?
Not always, though they often lead to polarity. In specific geometries like square planar (XeF₄) or linear (I₃⁻), lone pairs are positioned symmetrically so that their effects cancel out, resulting in a nonpolar species.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.