NMR Interpretation Practice Questions with Answers
Concept Explanation
NMR Interpretation is the process of analyzing Nuclear Magnetic Resonance spectra to determine the physical and chemical properties of atoms or the molecules in which they are contained. This analytical technique relies on the magnetic properties of certain atomic nuclei, most commonly the proton (1H) and carbon-13 (13C), to map out the connectivity and environment of atoms within a substance. By examining the chemical shift, integration, and multiplicity of signals, chemists can deduce the precise structure of complex organic compounds.
To master NMR Interpretation, one must understand four key pieces of information provided by a 1H NMR spectrum:
-
Number of Signals: This indicates how many sets of unique (non-equivalent) protons are in the molecule.
-
Chemical Shift (δ): Measured in parts per million (ppm), this tells us the electronic environment of the protons. Protons near electronegative atoms or pi systems are "deshielded" and appear at higher ppm.
-
Integration: The area under each peak corresponds to the relative number of protons contributing to that signal.
-
Splitting Pattern (Multiplicity): Following the n+1 rule, the number of peaks in a signal tells us how many neighboring protons are present on adjacent carbons.
Understanding these concepts often requires a solid foundation in other areas of structural chemistry, such as Lewis Structure Practice Questions to visualize atom connectivity. According to the IUPAC Gold Book, NMR is indispensable because it provides a non-destructive way to examine molecular dynamics and structure in solution.
Solved Examples
Below are three fully worked examples of identifying structures based on NMR data.
Example 1: Identifying a Simple Alkane
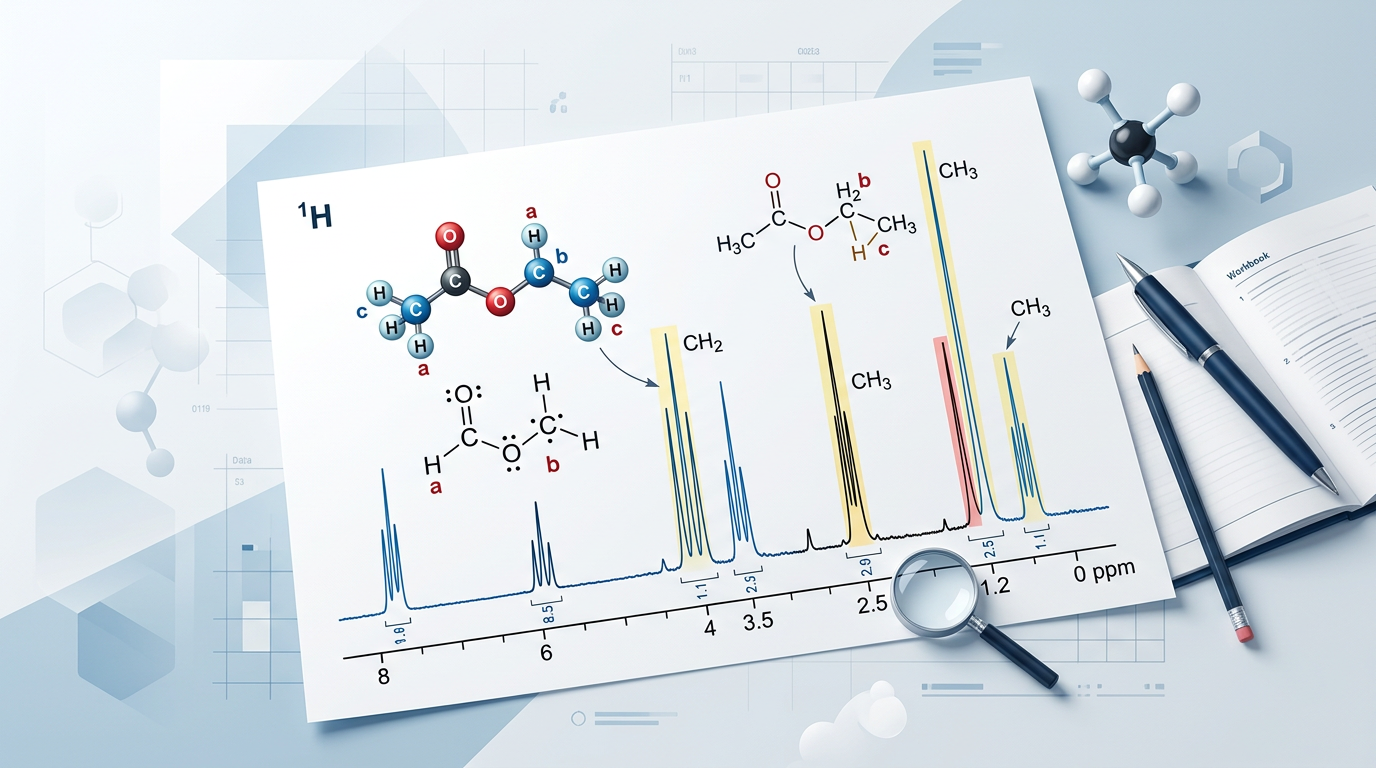
Problem: A compound with the formula C2H6O shows two signals in its 1H NMR spectrum: a triplet at 1.2 ppm (3H) and a quartet at 3.7 ppm (2H). A broad singlet is also seen at 2.5 ppm (1H). Identify the molecule.
-
Calculate Degrees of Unsaturation: (2C + 2 - H)/2 = (4+2-6)/2 = 0. The molecule is saturated.
-
Analyze the Multiplicity: A triplet (3H) and a quartet (2H) is the classic signature of an ethyl group (-CH2CH3). The quartet is at 3.7 ppm, which is significantly deshielded, suggesting the CH2 is attached to an oxygen.
-
Analyze the Singlet: The 1H singlet at 2.5 ppm is typical for an alcohol (-OH) group, which often doesn't show splitting due to rapid proton exchange.
-
Conclusion: The molecule is Ethanol (CH3CH2OH).
Example 2: Aromatic Substitution
Problem: A compound with formula C8H10 shows two signals: a singlet at 2.3 ppm (6H) and a singlet at 7.1 ppm (4H).
-
Degree of Unsaturation: (16 + 2 - 10)/2 = 4. This usually indicates a benzene ring.
-
Chemical Shifts: The signal at 7.1 ppm is in the aromatic region. Since it integrates to 4H, there are 4 protons on the ring, meaning the ring is disubstituted.
-
Symmetry: Both signals are singlets. For the 6H signal at 2.3 ppm to be a singlet, the two methyl groups (2 x CH3) must be equivalent and have no neighboring protons. For the 4H aromatic signal to be a singlet, all four ring protons must be chemically equivalent.
-
Conclusion: This symmetry only exists in p-xylene (1,4-dimethylbenzene).
Example 3: Carbonyl Environment
Problem: C3H6O shows a single sharp singlet at 2.1 ppm.
-
Degree of Unsaturation: (6 + 2 - 6)/2 = 1. Likely a double bond (C=O or C=C).
-
Chemical Shift: 2.1 ppm is typical for protons alpha to a carbonyl group (CH3-C=O).
-
Integration/Symmetry: Since all 6 protons appear as one singlet, they must all be equivalent and have no neighbors.
-
Conclusion: Two equivalent methyl groups attached to a central carbonyl. The molecule is Acetone (CH3COCH3).
Practice Questions
Test your skills with these NMR interpretation practice questions. Use the chemical shift tables and the n+1 rule to deduce the structures.
1. A compound with molecular formula C4H8O2 shows a triplet at 1.2 ppm (3H), a singlet at 2.0 ppm (3H), and a quartet at 4.1 ppm (2H). What is the structure?
2. Identify the compound C3H7Cl that produces a doublet at 1.5 ppm (6H) and a septet at 4.2 ppm (1H).
3. A molecule has the formula C7H8. Its 1H NMR spectrum shows a singlet at 2.3 ppm (3H) and a multiplet at 7.2 ppm (5H). Name the compound.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. (Medium) A compound C5H10O shows a singlet at 1.1 ppm (9H) and a singlet at 2.1 ppm (1H). Wait, correction: the formula is C5H10O and the signals are a singlet at 1.1 ppm (9H) and a singlet at 9.7 ppm (1H). Identify the structure.
5. (Medium) An unknown compound C10H12O2 shows a triplet at 1.3 ppm (3H), a quartet at 4.3 ppm (2H), and two doublets in the aromatic region at 7.0 ppm (2H) and 7.8 ppm (2H). What is the substitution pattern?
6. (Hard) A compound C4H10O shows a doublet at 1.2 ppm (6H), a multiplet at 1.8 ppm (1H), and a doublet at 3.4 ppm (2H). A broad singlet is at 2.0 ppm (1H). Propose a structure.
7. (Hard) Interpret the 1H NMR of C4H7ClO: triplet at 1.1 ppm (3H), quartet at 2.1 ppm (2H), and triplet at 4.5 ppm (2H).
8. (Hard) A compound C6H12 shows only one singlet in its 1H NMR spectrum. What is the most likely structure?
Answers & Explanations
-
Ethyl Acetate: The triplet (1.2 ppm) and quartet (4.1 ppm) indicate an ethyl group. The 4.1 ppm shift for the CH2 suggests it is attached to an ester oxygen (-O-CH2CH3). The singlet at 2.0 ppm is a methyl group next to a carbonyl (CH3CO-).
-
2-Chloropropane (Isopropyl chloride): The doublet (6H) indicates two equivalent methyl groups adjacent to a single proton (CH). The septet (1H) at 4.2 ppm confirms the CH is coupled to 6 neighbors and is deshielded by the Chlorine atom.
-
Toluene: The 5H multiplet at 7.2 ppm is characteristic of a monosubstituted benzene ring. The 3H singlet at 2.3 ppm is a methyl group attached directly to the ring (benzylic protons).
-
Pivalaldehyde (2,2-dimethylpropanal): The 9H singlet at 1.1 ppm represents a tert-butyl group (three equivalent methyls with no neighbors). The 1H singlet at 9.7 ppm is the highly deshielded aldehyde proton.
-
Ethyl 4-hydroxybenzoate (or similar para-substituted ester): The ethyl group is clear (triplet/quartet). The two aromatic doublets (2H each) indicate a 1,4-disubstituted (para) benzene ring where the two sides of the ring are chemically different.
-
Isobutanol (2-methyl-1-propanol): The 6H doublet and 1H multiplet indicate an isopropyl group (CH3)2CH-. The 2H doublet at 3.4 ppm is a CH2 group attached to the isopropyl and an OH group.
-
1-chlorobutan-2-one: (Correction based on shifts) The 1.1 (t, 3H) and 2.1 (q, 2H) indicate an ethyl group. The 4.5 (t, 2H) is a CH2 next to a Cl and a carbonyl. A common structure fitting this is 1-chloro-2-butanone.
-
Cyclohexane: In cyclohexane, all 12 protons are chemically equivalent due to rapid chair-chair interconversion at room temperature, resulting in a single singlet.
For more practice on related electronic effects, check out our Periodic Trends Practice Questions or explore Hybridization Practice Questions with Answers to better understand orbital overlap in these molecules.
Quick Quiz
1. What does the n+1 rule predict in 1H NMR?
- A The number of carbon atoms
- B The number of peaks in a signal based on neighboring protons
- C The exact chemical shift of a proton
- D The total number of protons in the molecule
Check answer
Answer: B. The number of peaks in a signal based on neighboring protons
2. Which of the following protons would appear furthest downfield (highest ppm)?
- A CH4 (Methane)
- B CH3Cl (Chloromethane)
- C CH2Cl2 (Dichloromethane)
- D CHCl3 (Chloroform)
- E CH3OH (Methanol)
Check answer
Answer: D. CHCl3 (Chloroform)
3. An integration value of 3:2 in a spectrum of C5H10O likely represents:
- A 3 carbons and 2 oxygens
- B A ratio of 6 protons to 4 protons
- C A methyl group and a hydroxyl group
- D The distance between two peaks
Check answer
Answer: B. A ratio of 6 protons to 4 protons
4. Where would you typically find the signal for an aromatic proton?
- A 0.5 - 1.5 ppm
- B 2.0 - 3.0 ppm
- C 6.5 - 8.5 ppm
- D 9.0 - 10.0 ppm
Check answer
Answer: C. 6.5 - 8.5 ppm
5. Why does TMS (Tetramethylsilane) serve as a standard in NMR?
- A It is highly reactive
- B Its protons are highly deshielded
- C It provides a single peak at 0 ppm that is easily removed
- D It has 4 different types of protons
Check answer
Answer: C. It provides a single peak at 0 ppm that is easily removed
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the chemical shift in NMR?
The chemical shift is the resonant frequency of a nucleus relative to a standard (usually TMS), expressed in parts per million (ppm). it reflects the electronic environment and shielding levels around a specific nucleus.
How do neighboring protons affect the NMR signal?
Neighboring protons cause spin-spin coupling, which splits the signal into multiple peaks according to the n+1 rule. This allows chemists to determine how many hydrogens are attached to adjacent carbon atoms.
Why do some signals appear as broad singlets?
Broad singlets often occur for protons attached to oxygen (OH) or nitrogen (NH) due to rapid intermolecular proton exchange. This exchange blurs the coupling effects that would otherwise cause splitting.
What is the difference between shielding and deshielding?
Shielding occurs when electron density around a nucleus opposes the external magnetic field, moving the signal upfield (lower ppm). Deshielding occurs when electronegative atoms pull electrons away, moving the signal downfield (higher ppm).
Can NMR distinguish between isomers?
Yes, NMR is one of the most powerful tools for distinguishing isomers because it reveals the specific connectivity and symmetry of a molecule. Even molecules with the same formula will produce distinct splitting patterns and shifts based on their unique structures.
What is the significance of integration in 1H NMR?
Integration represents the area under a peak, which is directly proportional to the number of hydrogen atoms causing that signal. By comparing integration ratios, you can determine the relative quantity of each type of proton in the molecule.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.