Easy Polarity Determination Practice Questions
Concept Explanation
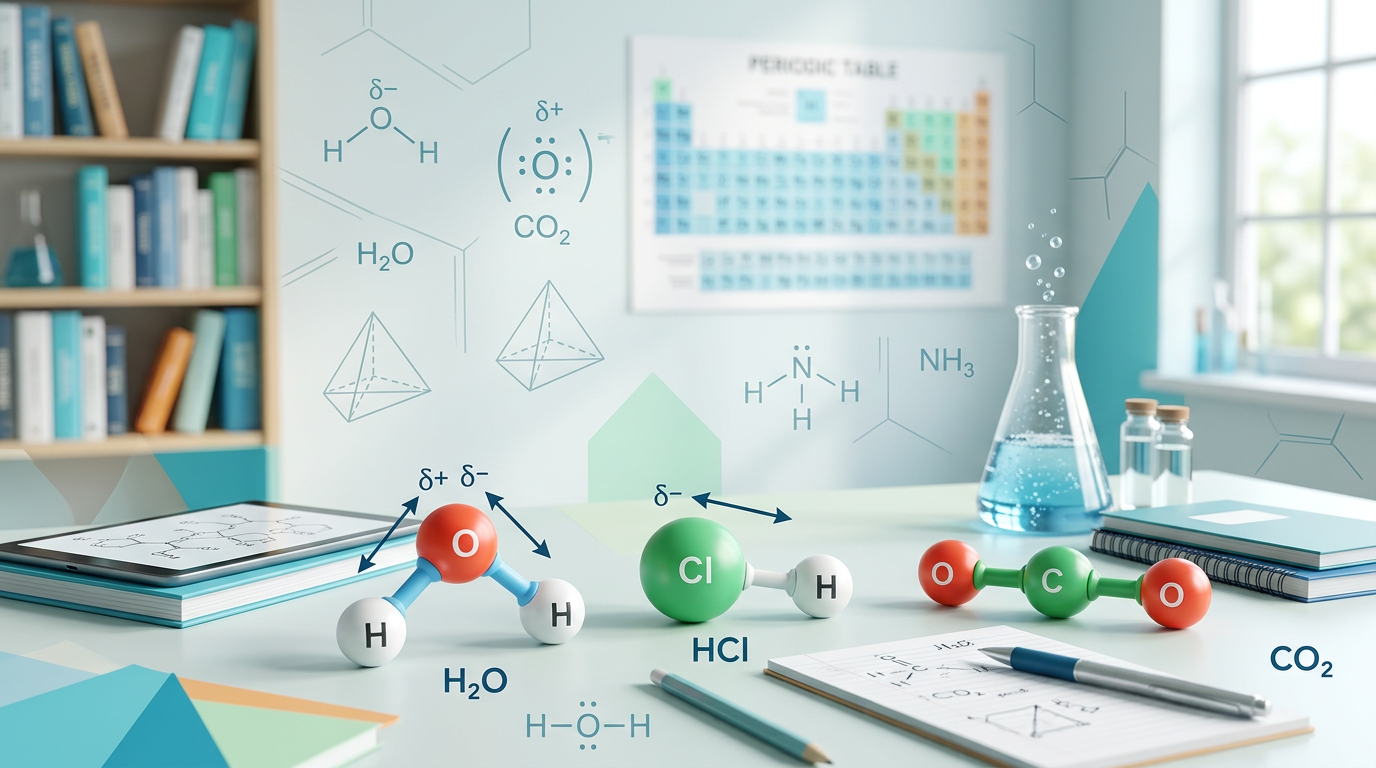
Polarity determination is the process of identifying whether a molecule has a net dipole moment based on the distribution of electrical charge across its atoms. This fundamental chemical property depends on two primary factors: bond polarity and molecular geometry. When atoms with different electronegativities share electrons, the electrons spend more time near the more electronegative atom, creating a polar covalent bond. However, a molecule containing polar bonds is not necessarily polar overall. According to Wikipedia's overview of chemical polarity, the symmetry of the molecule determines if these individual bond dipoles cancel each other out or add together to create a molecular dipole.
To master easy polarity determination, you must first understand periodic trends, specifically electronegativity. Electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. If the electronegativity difference between two atoms is between 0.5 and 1.7, the bond is typically considered polar. Once the bonds are identified, we use VSEPR geometry to see how those bonds are arranged in 3D space. Highly symmetric shapes like linear (with identical ends), trigonal planar, and tetrahedral often result in nonpolar molecules because the dipoles cancel. Conversely, asymmetric shapes or molecules with lone pairs on the central atom, like bent or trigonal pyramidal, almost always result in polar molecules.
A simple rule of thumb for beginners is to look for "symmetry breakers." A molecule is likely polar if it has lone pairs on the central atom (with a few rare exceptions) or if the atoms attached to the central atom are not all the same. For example, CH4 is nonpolar because the four C-H bonds are arranged symmetrically, but CH3Cl is polar because the C-Cl bond is different from the others, creating an uneven pull.
Solved Examples
Below are three step-by-step examples demonstrating how to determine molecular polarity using electronegativity and geometry.
-
Determine if Carbon Dioxide (CO2) is polar or nonpolar.
-
Identify the bonds: Carbon and Oxygen have an electronegativity difference (3.5 - 2.5 = 1.0), so the C=O bonds are polar.
-
Determine the geometry: CO2 has a linear molecular geometry (O=C=O).
-
Evaluate symmetry: The two polar bonds point in exactly opposite directions (180 degrees apart).
-
Conclusion: The bond dipoles cancel out perfectly, making CO2 a nonpolar molecule.
-
-
Determine if Water (H2O) is polar or nonpolar.
-
Identify the bonds: Oxygen (3.5) and Hydrogen (2.1) have a difference of 1.4, indicating polar O-H bonds.
-
Determine the geometry: Due to two lone pairs on the oxygen, the shape is bent (approx. 104.5 degrees).
-
Evaluate symmetry: The dipoles do not cancel because they are pointing toward the oxygen at an angle, creating a net partial negative charge near the oxygen and a partial positive charge near the hydrogens.
-
Conclusion: Water is a polar molecule.
-
-
Determine if Boron Trifluoride (BF3) is polar or nonpolar.
-
Identify the bonds: B-F bonds are highly polar (4.0 - 2.0 = 2.0 difference).
-
Determine the geometry: Boron has no lone pairs and three bonding pairs, resulting in a trigonal planar shape.
-
Evaluate symmetry: The three identical B-F dipoles are spread 120 degrees apart in a single plane, cancelling each other out.
-
Conclusion: BF3 is nonpolar.
-
Practice Questions
Test your skills with these easy polarity determination practice questions. Remember to draw the Lewis structure if you are unsure of the geometry.
1. Is the diatomic molecule HCl polar or nonpolar?
2. Determine the polarity of Methane (CH4).
3. Consider Ammonia (NH3). Does it have a net molecular dipole?
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. Is Nitrogen gas (N2) polar or nonpolar?
5. Determine the polarity of Sulfur Dioxide (SO2).
6. Is Carbon Tetrachloride (CCl4) a polar molecule?
7. Determine the polarity of Hydrogen Sulfide (H2S).
8. Is the molecule Chloromethane (CH3Cl) polar or nonpolar?
9. Determine if Phosphorus Trichloride (PCl3) is polar.
10. Is Beryllium Chloride (BeCl2) polar or nonpolar?
Answers & Explanations
-
1. Polar: Since HCl is a diatomic molecule with two different atoms, the electronegativity difference between H (2.1) and Cl (3.0) creates a permanent dipole. There is no other bond to cancel it out.
-
2. Nonpolar: CH4 has a tetrahedral geometry. Because all four surrounding atoms are hydrogen, the small bond dipoles cancel out perfectly due to the high symmetry of the molecule.
-
3. Polar: NH3 has a trigonal pyramidal shape because of the lone pair on the nitrogen. This asymmetry prevents the N-H bond dipoles from cancelling, resulting in a net dipole moment.
-
4. Nonpolar: N2 consists of two identical nitrogen atoms. Since they have the same electronegativity, the electrons are shared equally, and there is no bond dipole.
-
5. Polar: SO2 has a bent molecular geometry due to the lone pair on the sulfur atom. The polar S-O bonds do not cancel out in this V-shape.
-
6. Nonpolar: Similar to methane, CCl4 is tetrahedral. Since all four atoms attached to the carbon are chlorine, the dipoles cancel out despite the C-Cl bonds being individually polar.
-
7. Polar: H2S has a bent geometry similar to water. Sulfur is more electronegative than hydrogen, and the bent shape ensures the dipoles do not cancel.
-
8. Polar: While the geometry is tetrahedral, the four surrounding atoms are not identical (three H and one Cl). The C-Cl bond is much more polar than the C-H bonds, creating an imbalance.
-
9. Polar: PCl3 has a trigonal pyramidal geometry (one lone pair on P). The three polar P-Cl bonds point away from the lone pair, leading to a net molecular dipole.
-
10. Nonpolar: BeCl2 is a linear molecule. The two Be-Cl bond dipoles point in opposite directions and cancel each other out.
Quick Quiz
1. Which of the following conditions is required for a molecule to be nonpolar?
- A It must contain only nonpolar bonds
- B The bond dipoles must cancel out due to symmetry
- C It must have at least one lone pair on the central atom
- D It must consist of more than three atoms
Check answer
Answer: B. The bond dipoles must cancel out due to symmetry
2. Why is water (H2O) a polar molecule?
- A It has a linear shape that pulls electrons to one side
- B It has a bent geometry that prevents bond dipoles from cancelling
- C Oxygen and Hydrogen have the same electronegativity
- D The hydrogen atoms are more electronegative than oxygen
Check answer
Answer: B. It has a bent geometry that prevents bond dipoles from cancelling
3. Which molecule is nonpolar despite having polar bonds?
- A NH3
- B H2O
- C CCl4
- D HCl
Check answer
Answer: C. CCl4
4. What happens to the polarity of a tetrahedral molecule if one of the four identical outer atoms is replaced by a different atom?
- A It becomes polar
- B It remains nonpolar
- C It becomes an ion
- D The bond angles increase to 180 degrees
Check answer
Answer: A. It becomes polar
5. Which of these shapes is most likely to result in a polar molecule if the outer atoms are identical?
- A Linear
- B Trigonal Planar
- C Tetrahedral
- D Trigonal Pyramidal
Check answer
Answer: D. Trigonal Pyramidal
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the difference between a polar bond and a polar molecule?
A polar bond is a local attraction of electrons between two specific atoms, whereas a polar molecule is the sum of all bond dipoles in 3D space. You can have polar bonds in a nonpolar molecule if the geometry is symmetric enough to cancel the dipoles out.
Can a molecule with lone pairs be nonpolar?
Yes, though it is rare in introductory chemistry; molecules like XeF4 are nonpolar because their square planar geometry allows the lone pairs and bond dipoles to cancel. For most simple molecules, however, a lone pair on the central atom usually indicates polarity.
How does electronegativity affect polarity?
Electronegativity determines how strongly an atom attracts electrons in a bond, creating partial charges. The larger the difference in electronegativity between two bonded atoms, the more polar that specific bond becomes.
Is CO2 polar or nonpolar?
CO2 is nonpolar because its linear geometry causes the two equal and opposite C=O bond dipoles to cancel each other out. This is a classic example of how molecular shape overrides bond polarity.
Why is molecular geometry important for polarity?
Geometry determines the spatial orientation of bond dipoles, which are vector quantities. If the vectors are arranged symmetrically, they sum to zero, but if they are asymmetrical, they create a net molecular dipole.
Does a nonpolar molecule have any charged ends?
No, a nonpolar molecule has a symmetric distribution of charge, meaning it lacks a permanent positive or negative pole. While it may have temporary fluctuations, it does not have a constant dipole moment like polar molecules do.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.