Charles’s Law Practice Questions with Answers
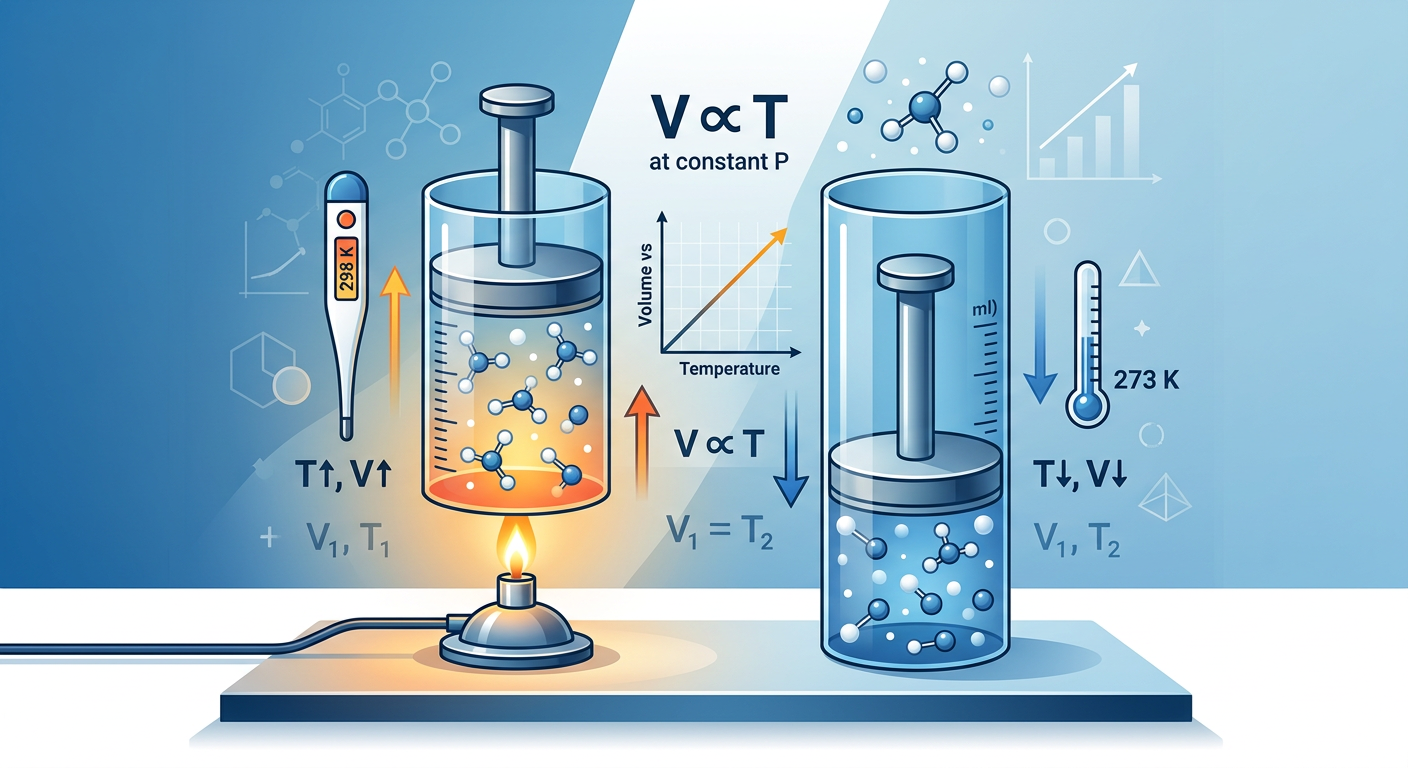
Concept Explanation
Charles’s Law states that the volume of a given mass of gas is directly proportional to its absolute temperature, provided the pressure remains constant. This fundamental principle of thermodynamics was first observed by Jacques Charles in the 1780s and later popularized by Joseph Louis Gay-Lussac. In simpler terms, as a gas gets hotter, it expands; as it cools, it contracts. For students who need to study for exams in engineering school or chemistry, understanding this relationship is essential for mastering gas behavior.
The mathematical representation of Charles’s Law is expressed as:
V₁ / T₁ = V₂ / T₂
Where:
-
V₁ is the initial volume.
-
T₁ is the initial absolute temperature.
-
V₂ is the final volume.
-
T₂ is the final absolute temperature.
A critical requirement for using this formula is that temperature must be expressed in Kelvins (K), not Celsius (°C) or Fahrenheit (°F). Since the Kelvin scale starts at absolute zero, it ensures that the mathematical ratio remains consistent. To convert from Celsius to Kelvin, you simply add 273.15 to the Celsius temperature (though 273 is often used in basic practice). If you find yourself struggling to study for exams when overwhelmed by different gas laws, remember that Charles’s Law focuses exclusively on the relationship between Volume and Temperature.
The Kinetic Molecular Theory Perspective
According to the Kinetic Molecular Theory, gas particles are in constant, random motion. When the temperature of a gas increases, the average kinetic energy of the particles increases. These faster-moving particles collide with the walls of their container more frequently and with greater force. To keep the internal pressure constant against the external pressure, the volume of the container must increase to spread out these collisions.
Solved Examples
These examples demonstrate how to apply the formula to real-world scenarios. Following these steps can help if you are learning how to study for exams to get straight A’s in science subjects.
Example 1: Finding Final Volume
A balloon is filled with 2.50 L of air at a temperature of 25°C. If the balloon is placed in a freezer where the temperature is -10°C, what will be the new volume of the balloon, assuming pressure remains constant?
-
Identify the variables: V₁ = 2.50 L, T₁ = 25°C, T₂ = -10°C.
-
Convert temperatures to Kelvin: T₁ = 25 + 273 = 298 K; T₂ = -10 + 273 = 263 K.
-
Set up the equation: V₁ / T₁ = V₂ / T₂ → 2.50 / 298 = V₂ / 263.
-
Solve for V₂: V₂ = (2.50 × 263) / 298.
-
Calculation: V₂ ≈ 2.21 L.
Example 2: Finding Initial Temperature
A sample of nitrogen gas occupies a volume of 500 mL at an unknown temperature. When the gas is heated to 100°C, the volume increases to 750 mL. What was the initial temperature in Celsius?
-
Identify the variables: V₁ = 500 mL, V₂ = 750 mL, T₂ = 100°C.
-
Convert T₂ to Kelvin: T₂ = 100 + 273 = 373 K.
-
Set up the equation: 500 / T₁ = 750 / 373.
-
Solve for T₁: T₁ = (500 × 373) / 750 = 248.67 K.
-
Convert back to Celsius: T₁ = 248.67 - 273 = -24.33°C.
Example 3: Volume Change in a Cylinder
A piston-cylinder contains 1.2 L of gas at 300 K. If the volume needs to be increased to 1.8 L while keeping pressure constant, what temperature is required?
-
Identify the variables: V₁ = 1.2 L, T₁ = 300 K, V₂ = 1.8 L.
-
Set up the equation: 1.2 / 300 = 1.8 / T₂.
-
Solve for T₂: T₂ = (1.8 × 300) / 1.2.
-
Calculation: T₂ = 450 K.
Practice Questions
1. A sample of oxygen gas has a volume of 150 mL at 27°C. What volume will it occupy at 127°C, assuming constant pressure?
2. A rigid-walled container is not used here because Charles's Law requires volume to be able to change. Instead, imagine a flexible container with 4.0 L of gas at 200 K. If the temperature is doubled to 400 K, what is the new volume?
3. A gas occupies 3.5 L at 298 K. At what temperature (in Kelvin) will the volume reach 5.0 L?
Start Learning Smarter Today
Join thousands of students using AI-powered study tools to achieve better results.
Get Started Free4. If a gas occupies 12.0 L at 50°C, what will be its volume at 0°C?
5. A weather balloon contains 100 m³ of helium at 20°C. As it rises, the temperature drops to -40°C. If the pressure were to remain constant, what would the new volume be?
6. A sample of neon gas at 350 K has a volume of 0.75 L. To what temperature must the gas be cooled to reduce the volume to 0.50 L?
7. A 2.0 L sample of air at -50°C is heated until the volume is 3.0 L. What is the final temperature in Celsius?
8. A syringe contains 50 mL of gas at 22°C. The syringe is placed in boiling water (100°C). What is the new volume of the gas?
9. A gas occupies 250 mL at 30°C. If the volume increases to 400 mL, what is the new temperature in Kelvin?
10. If the volume of a gas at 546 K is 10 L, what was the volume at 273 K (standard temperature)?
Answers & Explanations
-
200 mL: Convert T₁ (27°C = 300 K) and T₂ (127°C = 400 K). V₂ = (150 mL × 400 K) / 300 K = 200 mL.
-
8.0 L: Charles’s Law is a direct proportion. If the absolute temperature doubles (from 200 K to 400 K), the volume must also double. 4.0 L × 2 = 8.0 L.
-
425.7 K: T₂ = (V₂ × T₁) / V₁ = (5.0 L × 298 K) / 3.5 L = 425.7 K.
-
10.14 L: Convert T₁ (50°C = 323 K) and T₂ (0°C = 273 K). V₂ = (12.0 L × 273 K) / 323 K = 10.14 L.
-
79.52 m³: Convert T₁ (20°C = 293 K) and T₂ (-40°C = 233 K). V₂ = (100 m³ × 233 K) / 293 K = 79.52 m³.
-
233.3 K: T₂ = (V₂ × T₁) / V₁ = (0.50 L × 350 K) / 0.75 L = 233.3 K.
-
61.5°C: Convert T₁ (-50°C = 223 K). T₂ = (3.0 L × 223 K) / 2.0 L = 334.5 K. Convert to Celsius: 334.5 - 273 = 61.5°C.
-
63.22 mL: Convert T₁ (22°C = 295 K) and T₂ (100°C = 373 K). V₂ = (50 mL × 373 K) / 295 K = 63.22 mL.
-
484.8 K: T₂ = (V₂ × T₁) / V₁ = (400 mL × 303 K) / 250 mL = 484.8 K.
-
5 L: T₁ = 546 K, V₁ = 10 L, T₂ = 273 K. V₂ = (10 L × 273 K) / 546 K = 5 L. Note that 273 K is exactly half of 546 K, so the volume halves.
Quick Quiz
1. Which of the following variables must remain constant for Charles’s Law to apply?
- A Volume
- B Temperature
- C Pressure
- D Mass and Pressure
Check answer
Answer: D. Mass and Pressure
2. What is the equivalent of 25°C in the Kelvin scale?
- A 25 K
- B 248.15 K
- C 298.15 K
- D 315.15 K
Check answer
Answer: C. 298.15 K
3. If the absolute temperature of a gas is tripled while pressure is constant, what happens to the volume?
- A It stays the same
- B It decreases by one-third
- C It triples
- D It increases by nine times
Check answer
Answer: C. It triples
4. Charles’s Law describes the relationship between which two properties?
- A Pressure and Volume
- B Volume and Temperature
- C Pressure and Temperature
- D Mass and Volume
Check answer
Answer: B. Volume and Temperature
5. A graph of Volume (y-axis) vs. Temperature in Kelvin (x-axis) for a gas at constant pressure would look like:
- A A downward curving line
- B A horizontal line
- C A straight line passing through the origin
- D A vertical line
Check answer
Answer: C. A straight line passing through the origin
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the formula for Charles’s Law?
The formula for Charles’s Law is V₁ / T₁ = V₂ / T₂, where V represents volume and T represents absolute temperature in Kelvin. This equation allows you to calculate the change in one variable when the other changes, provided pressure and amount of gas remain constant.
Why must temperature be in Kelvin for Charles’s Law?
Temperature must be in Kelvin because the law relies on a direct mathematical proportion starting from absolute zero. Using Celsius or Fahrenheit, which have arbitrary zero points and negative values, would result in incorrect or impossible ratios (such as negative volumes).
Does Charles’s Law apply to liquids?
No, Charles’s Law specifically describes the behavior of gases because gas particles have significant space between them, allowing for large changes in volume with temperature. Liquids and solids are relatively incompressible and do not expand or contract significantly enough to follow this simple linear relationship.
What happens to a gas at absolute zero according to Charles’s Law?
Theoretically, according to the mathematical model of Charles’s Law, the volume of a gas would become zero at absolute zero (0 K). However, in reality, all gases liquefy or solidify before reaching that temperature, and the ideal gas assumptions no longer apply.
How does Charles’s Law relate to hot air balloons?
Hot air balloons operate on Charles’s Law: as the air inside the balloon is heated, its volume increases and its density decreases compared to the cooler air outside. This lower density provides the buoyancy needed for the balloon to lift off the ground.
What is the difference between Charles’s Law and Boyle’s Law?
The main difference is the variables they relate: Charles’s Law relates Volume and Temperature at constant Pressure, while Boyle’s Law relates Volume and Pressure at constant Temperature. They are both components of the Combined Gas Law used in general chemistry.
Start Learning Smarter Today
Join thousands of students using AI-powered study tools to achieve better results.
Get Started FreeEnjoyed this article?
Share it with others who might find it helpful.