Medium Hess’s Law Practice Questions
Concept Explanation
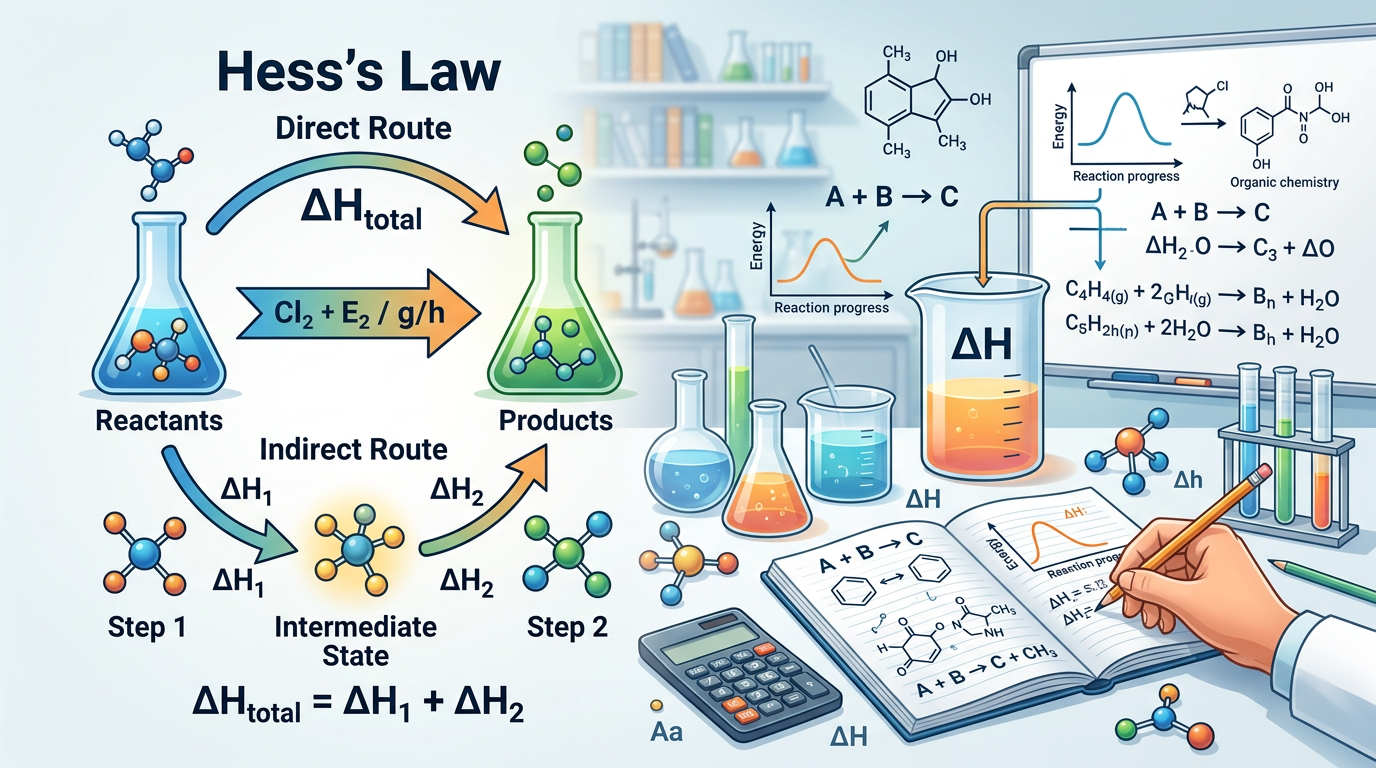
Hess’s Law states that the total enthalpy change for a chemical reaction is the same regardless of the path taken, provided the initial and final states are identical. This fundamental principle of thermochemistry is a direct application of the law of conservation of energy and the fact that enthalpy is a state function. When you are solving Medium Hess’s Law Practice Questions, you must manipulate a series of intermediate chemical equations so that they sum up to a target equation. If you reverse a reaction, the sign of its enthalpy change (ΔH) must be flipped; if you multiply the coefficients of a reaction by a factor, you must multiply the ΔH by that same factor. This method is particularly useful for calculating the heat of reaction for processes that are difficult to measure directly in a lab setting, such as the formation of carbon monoxide from solid carbon. According to the International Union of Pure and Applied Chemistry (IUPAC), standard enthalpy changes are typically reported at 298.15 K and 1 bar of pressure.
Solved Examples
-
Calculate the ΔH for the reaction: C(s) + 1/2 O₂(g) → CO(g)
Given:
(1) C(s) + O₂(g) → CO₂(g) ΔH = -393.5 kJ
(2) CO(g) + 1/2 O₂(g) → CO₂(g) ΔH = -283.0 kJ
Solution:-
We need CO(g) as a product, but it is a reactant in equation (2). Reverse equation (2): CO₂(g) → CO(g) + 1/2 O₂(g) ΔH = +283.0 kJ.
-
Keep equation (1) as is: C(s) + O₂(g) → CO₂(g) ΔH = -393.5 kJ.
-
Add the equations: C(s) + O₂(g) + CO₂(g) → CO₂(g) + CO(g) + 1/2 O₂(g).
-
Cancel common terms (CO₂ and 1/2 O₂): C(s) + 1/2 O₂(g) → CO(g).
-
Sum the ΔH values: -393.5 kJ + 283.0 kJ = -110.5 kJ.
-
-
Find ΔH for: 2 NO₂(g) → N₂O₄(g)
Given:
(1) N₂(g) + 2 O₂(g) → 2 NO₂(g) ΔH = +67.7 kJ
(2) N₂(g) + 2 O₂(g) → N₂O₄(g) ΔH = +9.7 kJ
Solution:-
Reverse equation (1) to get 2 NO₂ on the reactant side: 2 NO₂(g) → N₂(g) + 2 O₂(g) ΔH = -67.7 kJ.
-
Keep equation (2) as is: N₂(g) + 2 O₂(g) → N₂O₄(g) ΔH = +9.7 kJ.
-
Add the equations: 2 NO₂(g) + N₂(g) + 2 O₂(g) → N₂(g) + 2 O₂(g) + N₂O₄(g).
-
Cancel common terms: 2 NO₂(g) → N₂O₄(g).
-
Sum the ΔH: -67.7 kJ + 9.7 kJ = -58.0 kJ.
-
-
Determine ΔH for: 2C(s) + H₂(g) → C₂H₂(g)
Given:
(1) C₂H₂(g) + 5/2 O₂(g) → 2CO₂(g) + H₂O(l) ΔH = -1299.5 kJ
(2) C(s) + O₂(g) → CO₂(g) ΔH = -393.5 kJ
(3) H₂(g) + 1/2 O₂(g) → H₂O(l) ΔH = -285.8 kJ
Solution:-
Multiply equation (2) by 2 to get 2C(s): 2C(s) + 2O₂(g) → 2CO₂(g) ΔH = 2(-393.5) = -787.0 kJ.
-
Keep equation (3) as is: H₂(g) + 1/2 O₂(g) → H₂O(l) ΔH = -285.8 kJ.
-
Reverse equation (1) to get C₂H₂(g) on the product side: 2CO₂(g) + H₂O(l) → C₂H₂(g) + 5/2 O₂(g) ΔH = +1299.5 kJ.
-
Sum the reactions and enthalpies: -787.0 - 285.8 + 1299.5 = +226.7 kJ.
-
Practice Questions
1. Calculate the enthalpy change for the reaction: P₄O₆(s) + 2 O₂(g) → P₄O₁₀(s).
Given:
(1) P₄(s) + 3 O₂(g) → P₄O₆(s) ΔH = -1640.1 kJ
(2) P₄(s) + 5 O₂(g) → P₄O₁₀(s) ΔH = -2940.1 kJ
2. Find the ΔH for the following reaction: WO₃(s) + 3 H₂(g) → W(s) + 3 H₂O(g).
Given:
(1) 2 W(s) + 3 O₂(g) → 2 WO₃(s) ΔH = -1685.4 kJ
(2) 2 H₂(g) + O₂(g) → 2 H₂O(g) ΔH = -477.8 kJ
3. Determine ΔH for the reaction: 2 S(s) + 3 O₂(g) → 2 SO₃(g).
Given:
(1) S(s) + O₂(g) → SO₂(g) ΔH = -296.8 kJ
(2) 2 SO₂(g) + O₂(g) → 2 SO₃(g) ΔH = -197.8 kJ
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. Calculate the enthalpy of formation of methane (CH₄): C(s) + 2 H₂(g) → CH₄(g).
Given:
(1) C(s) + O₂(g) → CO₂(g) ΔH = -393.5 kJ
(2) H₂(g) + 1/2 O₂(g) → H₂O(l) ΔH = -285.8 kJ
(3) CH₄(g) + 2 O₂(g) → CO₂(g) + 2 H₂O(l) ΔH = -890.3 kJ
5. Find ΔH for the reaction: ClF(g) + F₂(g) → ClF₃(g).
Given:
(1) 2 ClF(g) + O₂(g) → Cl₂O(g) + F₂O(g) ΔH = +167.4 kJ
(2) 2 ClF₃(g) + 2 O₂(g) → Cl₂O(g) + 3 F₂O(g) ΔH = +341.4 kJ
(3) 2 F₂(g) + O₂(g) → 2 F₂O(g) ΔH = -43.4 kJ
6. Calculate ΔH for: N₂H₄(l) + CH₄O(l) → CH₂O(g) + N₂(g) + 3 H₂(g).
Given:
(1) 2 NH₃(g) → N₂H₄(l) + H₂(g) ΔH = +22.5 kJ
(2) 2 NH₃(g) → N₂(g) + 3 H₂(g) ΔH = +57.5 kJ
(3) CH₂O(g) + H₂(g) → CH₄O(l) ΔH = -81.2 kJ
7. Determine the ΔH for: 4 NH₃(g) + 5 O₂(g) → 4 NO(g) + 6 H₂O(g).
Given:
(1) N₂(g) + O₂(g) → 2 NO(g) ΔH = +180.5 kJ
(2) N₂(g) + 3 H₂(g) → 2 NH₃(g) ΔH = -91.8 kJ
(3) 2 H₂(g) + O₂(g) → 2 H₂O(g) ΔH = -483.6 kJ
8. Solve for ΔH: C₂H₄(g) + H₂(g) → C₂H₆(g).
Given:
(1) C₂H₄(g) + 3 O₂(g) → 2 CO₂(g) + 2 H₂O(l) ΔH = -1411 kJ
(2) C₂H₆(g) + 7/2 O₂(g) → 2 CO₂(g) + 3 H₂O(l) ΔH = -1560 kJ
(3) H₂(g) + 1/2 O₂(g) → H₂O(l) ΔH = -286 kJ
Answers & Explanations
-
Answer: -1300.0 kJ.
Reverse (1): P₄O₆(s) → P₄(s) + 3 O₂(g) ΔH = +1640.1 kJ. Keep (2): P₄(s) + 5 O₂(g) → P₄O₁₀(s) ΔH = -2940.1 kJ. Sum: 1640.1 + (-2940.1) = -1300.0 kJ. -
Answer: +125.9 kJ.
Reverse (1) and divide by 2: WO₃(s) → W(s) + 1.5 O₂(g) ΔH = +842.7 kJ. Multiply (2) by 1.5: 3 H₂(g) + 1.5 O₂(g) → 3 H₂O(g) ΔH = -716.7 kJ. Sum: 842.7 - 716.7 = +126.0 kJ (rounding may vary). -
Answer: -791.4 kJ.
Multiply (1) by 2: 2 S(s) + 2 O₂(g) → 2 SO₂(g) ΔH = -593.6 kJ. Keep (2): 2 SO₂(g) + O₂(g) → 2 SO₃(g) ΔH = -197.8 kJ. Sum: -593.6 - 197.8 = -791.4 kJ. -
Answer: -74.8 kJ.
Keep (1): ΔH = -393.5. Multiply (2) by 2: ΔH = 2(-285.8) = -571.6. Reverse (3): ΔH = +890.3. Sum: -393.5 - 571.6 + 890.3 = -74.8 kJ. This is a classic enthalpy change calculation. -
Answer: -108.7 kJ.
Divide (1) by 2: ClF + 0.5 O₂ → 0.5 Cl₂O + 0.5 F₂O ΔH = +83.7. Reverse (2) and divide by 2: 0.5 Cl₂O + 1.5 F₂O → ClF₃ + O₂ ΔH = -170.7. Divide (3) by 2: F₂ + 0.5 O₂ → F₂O ΔH = -21.7. Sum: 83.7 - 170.7 - 21.7 = -108.7 kJ. -
Answer: +116.2 kJ.
Reverse (1): N₂H₄ + H₂ → 2 NH₃ ΔH = -22.5. Keep (2): 2 NH₃ → N₂ + 3 H₂ ΔH = +57.5. Reverse (3): CH₄O → CH₂O + H₂ ΔH = +81.2. Sum: -22.5 + 57.5 + 81.2 = +116.2 kJ. -
Answer: -906.2 kJ.
Multiply (1) by 2: 2 N₂ + 2 O₂ → 4 NO ΔH = +361.0. Reverse (2) and multiply by 2: 4 NH₃ → 2 N₂ + 6 H₂ ΔH = +183.6. Multiply (3) by 3: 6 H₂ + 3 O₂ → 6 H₂O ΔH = -1450.8. Sum: 361.0 + 183.6 - 1450.8 = -906.2 kJ. -
Answer: -137 kJ.
Keep (1): ΔH = -1411. Reverse (2): ΔH = +1560. Keep (3): ΔH = -286. Sum: -1411 + 1560 - 286 = -137 kJ.
Quick Quiz
1. If a reaction is reversed, what happens to the sign of its enthalpy change (ΔH)?
- A It remains the same
- B It becomes zero
- C The sign is flipped
- D The value is squared
Check answer
Answer: C. The sign is flipped
2. Which property of enthalpy allows Hess's Law to work?
- A Enthalpy is a state function
- B Enthalpy is an intensive property
- C Enthalpy depends on the reaction rate
- D Enthalpy is always negative for spontaneous reactions
Check answer
Answer: A. Enthalpy is a state function
3. What must you do to ΔH if you multiply all coefficients in a balanced equation by 3?
- A Divide ΔH by 3
- B Multiply ΔH by 3
- C Keep ΔH the same
- D Triple the temperature of the reaction
Check answer
Answer: B. Multiply ΔH by 3
4. In the context of Hess's Law, what is the enthalpy of formation for a pure element in its standard state?
- A 1.0 kJ/mol
- B -100 kJ/mol
- C 0 kJ/mol
- D It varies by element
Check answer
Answer: C. 0 kJ/mol
5. Which law is Hess's Law essentially a specific application of?
- A The Law of Definite Proportions
- B The First Law of Thermodynamics
- C Boyle's Law
- D Le Chatelier's Principle
Check answer
Answer: B. The First Law of Thermodynamics
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the main purpose of Hess’s Law?
The main purpose is to calculate the enthalpy change of a reaction that is difficult or impossible to measure directly in a laboratory. By using known enthalpy changes of related reactions, scientists can mathematically derive the energy change for the target process.
Does Hess’s Law apply to entropy and Gibbs free energy?
Yes, because entropy and Gibbs free energy are also state functions. You can use the same summation techniques to find ΔS or ΔG for a reaction, provided the intermediate steps are also state functions under the same conditions.
Why is enthalpy considered a state function?
Enthalpy is a state function because its value depends only on the current state of the system (pressure, temperature, and composition) and not on the history or the path taken to reach that state. This is why UC Davis ChemWiki and other academic resources emphasize it in thermochemistry.
Can Hess’s Law be used for reactions in different phases?
Yes, but you must account for the enthalpy of phase changes (like vaporization or fusion). If one reaction produces liquid water and another produces gaseous water, you must include the ΔH of vaporization to correctly sum the equations.
How do you handle coefficients when using Hess’s Law?
Coefficients act as multipliers for the ΔH value. If you need to double a reaction to match your target equation, you must also double the ΔH value reported for that reaction. This ensures that the energy change is proportional to the amount of matter involved.
What should I do if the intermediate reactions don't cancel out?
If the intermediate species do not cancel out, you may need to find an additional reaction to bridge the gap or re-evaluate the multipliers you applied. Every substance not present in the final target equation must be canceled out through the summation process.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.