Hard Boyle’s Law Practice Questions
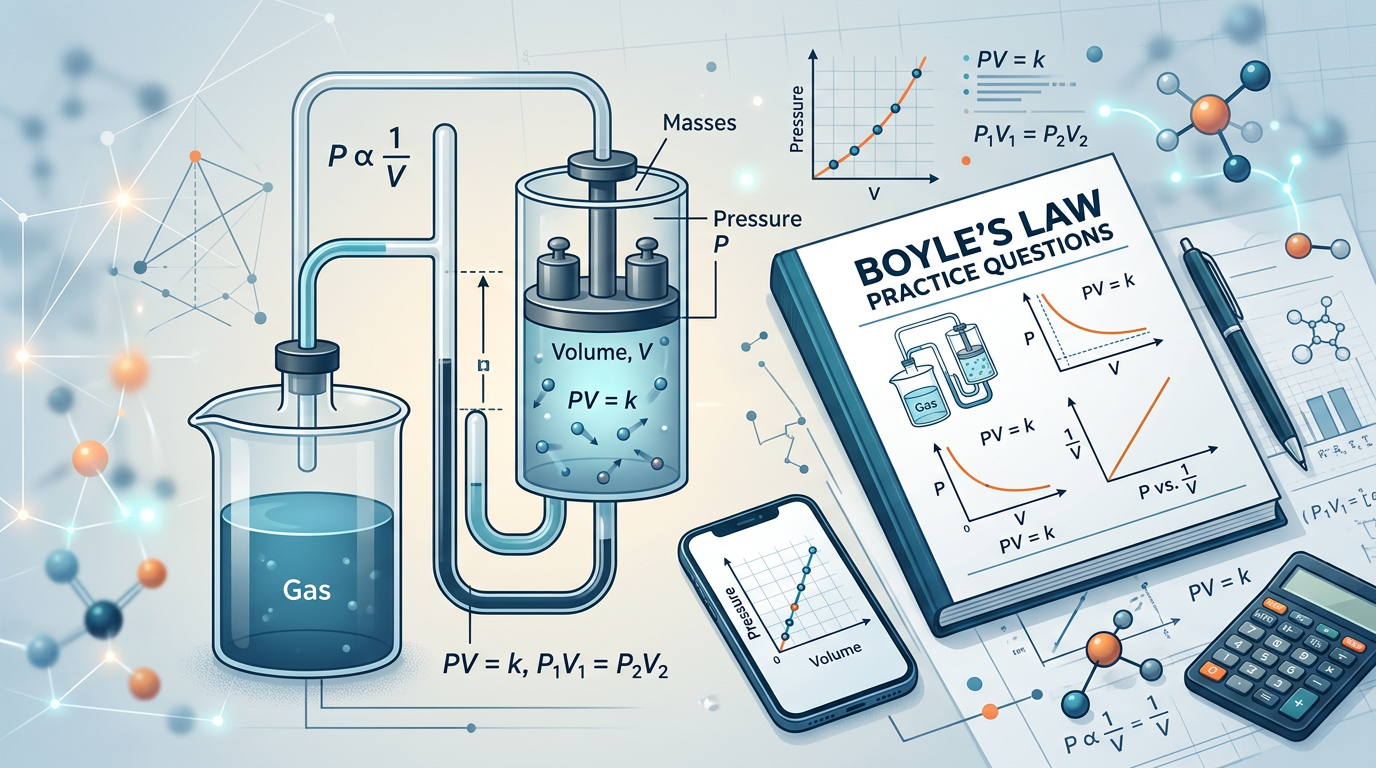
Concept Explanation
Boyle’s Law states that the pressure of a given mass of an ideal gas is inversely proportional to its volume when the temperature and amount of gas remain constant within a closed system.
This fundamental principle of gas mechanics was first published in 1662 by Robert Boyle. Mathematically, the law is expressed as P₁V₁ = P₂V₂, where P represents pressure and V represents volume. Because the relationship is inverse, if you decrease the volume of a container by half, the pressure exerted by the gas molecules inside will double, provided the temperature does not change. This happens because the same number of gas particles are now colliding with the container walls more frequently in a smaller space.
To master Hard Boyle’s Law Practice Questions, students must be comfortable with complex unit conversions and scenarios involving mercury manometers or changing atmospheric conditions. Unlike basic problems, advanced questions often require you to calculate the total pressure by accounting for both gauge pressure and atmospheric pressure. If you are preparing for rigorous exams, understanding how this law integrates with other gas properties is essential; for instance, you might eventually need to compare these results with calculations from Combined Gas Law Practice Questions.
Solved Examples
The following examples demonstrate how to solve complex gas law problems by breaking them down into logical steps.
Example 1: Solving for Final Pressure with Unit Conversion
A sample of nitrogen gas occupies a volume of 450 mL at a pressure of 1.2 atm. If the gas is compressed into a 0.15 L container at constant temperature, what is the final pressure in mmHg?
-
Identify knowns and unknowns: V₁ = 450 mL, P₁ = 1.2 atm, V₂ = 0.15 L, P₂ = ? (in mmHg).
-
Convert units to match: Convert V₂ to mL. 0.15 L × 1000 = 150 mL.
-
Apply the formula: P₁V₁ = P₂V₂ → (1.2 atm)(450 mL) = P₂(150 mL).
-
Calculate P₂ in atm: P₂ = (1.2 × 450) / 150 = 3.6 atm.
-
Convert final answer to mmHg: 3.6 atm × 760 mmHg/atm = 2,736 mmHg.
Example 2: The Piston Compression Challenge
A piston-cylinder assembly contains 2.5 m³ of air at a pressure of 101.3 kPa. The piston is moved until the pressure reaches 506.5 kPa. Calculate the change in volume (ΔV).
-
Identify variables: V₁ = 2.5 m³, P₁ = 101.3 kPa, P₂ = 506.5 kPa.
-
Set up the equation: (101.3 kPa)(2.5 m³) = (506.5 kPa)(V₂).
-
Solve for V₂: V₂ = (101.3 × 2.5) / 506.5 = 0.5 m³.
-
Determine the change: ΔV = V₂ - V₁ = 0.5 m³ - 2.5 m³ = -2.0 m³. (The volume decreased by 2.0 m³).
Example 3: Multi-Step Underwater Bubble Problem
A weather balloon is released from the bottom of a lake where the pressure is 3.0 atm. At the surface, the pressure is 1.0 atm. If the balloon's volume at the surface is 6.0 L, what was its volume at the bottom of the lake?
-
Identify variables: P₁ (bottom) = 3.0 atm, P₂ (surface) = 1.0 atm, V₂ (surface) = 6.0 L.
-
Set up the equation: (3.0 atm)(V₁) = (1.0 atm)(6.0 L).
-
Solve for V₁: V₁ = (1.0 × 6.0) / 3.0 = 2.0 L.
Practice Questions
1. A gas at 740 mmHg has a volume of 2.5 L. What will the pressure be if the volume is reduced to 1.8 L while keeping temperature constant?
2. A 5.0 L container of oxygen at 2.0 atm is connected by a valve to an empty 10.0 L container. When the valve is opened and the gas expands to fill both containers, what is the final pressure?
3. A deep-sea diver releases a 15 mL bubble of air at a depth where the pressure is 4.5 atm. What is the volume of the bubble when it reaches the surface where the pressure is 760 torr?
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →4. A sample of neon gas is held in a 0.75 L container at 150 kPa. If the gas is transferred to a container with a volume of 2,500 cm³, what is the new pressure in kPa?
5. A flexible container holds 45.0 L of gas at 1.05 atm. The container is compressed until the pressure is 45.0 psi. What is the final volume of the gas? (1 atm = 14.7 psi)
6. If a gas occupies 12.3 liters at a pressure of 0.985 atm, what is the volume when the pressure is increased to 1.50 × 10³ mmHg?
7. A gas cylinder with a volume of 20.0 L contains gas at a pressure of 2000 psi. If the gas is released into a room with a volume of 50.0 m³, what will the final pressure in the room be (assuming the room was previously a vacuum)?
8. A 3.5 L sample of gas at 1.0 atm is expanded to 7.0 L. Then, the pressure is tripled from that new state. What is the final volume?
9. A syringe contains 10.0 mL of air at 1.0 atm. The tip is sealed, and the plunger is pulled back to the 25.0 mL mark. What is the pressure inside the syringe in kPa?
10. An underwater bell contains 300 m³ of air at a depth where the pressure is 3.5 atm. If the bell is raised to a depth where the pressure is 1.2 atm, what is the new volume of air?
Answers & Explanations
1. 1,027.8 mmHg. Using P₁V₁ = P₂V₂, (740 mmHg)(2.5 L) = P₂(1.8 L). P₂ = 1850 / 1.8 = 1027.8 mmHg. Since volume decreased, pressure must increase.
2. 0.67 atm. The final volume (V₂) is the sum of both containers: 5.0 L + 10.0 L = 15.0 L. P₁V₁ = P₂V₂ → (2.0 atm)(5.0 L) = P₂(15.0 L). P₂ = 10 / 15 = 0.67 atm.
3. 67.5 mL. First, convert 760 torr to atm (760 torr = 1 atm). P₁V₁ = P₂V₂ → (4.5 atm)(15 mL) = (1 atm)(V₂). V₂ = 67.5 mL.
4. 45 kPa. Convert 2,500 cm³ to L. Since 1 cm³ = 1 mL, 2,500 cm³ = 2.5 L. (150 kPa)(0.75 L) = P₂(2.5 L). P₂ = 112.5 / 2.5 = 45 kPa.
5. 15.4 L. Convert P₂ to atm: 45.0 psi / 14.7 psi/atm = 3.06 atm. (1.05 atm)(45.0 L) = (3.06 atm)(V₂). V₂ = 47.25 / 3.06 = 15.44 L.
6. 6.14 L. Convert P₂ to atm: 1500 mmHg / 760 mmHg/atm = 1.974 atm. (0.985 atm)(12.3 L) = (1.974 atm)(V₂). V₂ = 12.1155 / 1.974 = 6.14 L.
7. 0.8 psi. Convert 50.0 m³ to liters: 50,000 L. (2000 psi)(20.0 L) = P₂(50,000 L). P₂ = 40,000 / 50,000 = 0.8 psi. This level of calculation is common in engineering school physics.
8. 2.33 L. Step 1: (1.0)(3.5) = P₂(7.0) → P₂ = 0.5 atm. Step 2: New pressure P₃ = 0.5 × 3 = 1.5 atm. New equation: (0.5 atm)(7.0 L) = (1.5 atm)(V₃). V₃ = 3.5 / 1.5 = 2.33 L.
9. 40.5 kPa. Convert 1.0 atm to kPa (101.325 kPa). (101.325 kPa)(10.0 mL) = P₂(25.0 mL). P₂ = 1013.25 / 25 = 40.53 kPa.
10. 875 m³. (3.5 atm)(300 m³) = (1.2 atm)(V₂). V₂ = 1050 / 1.2 = 875 m³.
Quick Quiz
1. If the pressure of a gas is tripled while temperature is held constant, what happens to the volume?
- A It triples
- B It remains the same
- C It is reduced to one-third
- D It is reduced to nine-tenths
Check answer
Answer: C. It is reduced to one-third
2. Which of the following units is NOT used to measure gas pressure?
- A Kelvin
- B Pascal
- C Atmosphere
- D Torr
Check answer
Answer: A. Kelvin
3. A 2.0 L balloon at 1.0 atm is submerged until the pressure is 4.0 atm. What is the new volume?
- A 8.0 L
- B 0.5 L
- C 1.0 L
- D 4.0 L
Check answer
Answer: B. 0.5 L
4. In the equation P₁V₁ = P₂V₂, what must remain constant for the law to apply?
- A Volume and Pressure
- B Temperature and Moles
- C Pressure and Temperature
- D Volume and Moles
Check answer
Answer: B. Temperature and Moles
5. A graph of Pressure (y-axis) vs. Volume (x-axis) for Boyle's Law results in what shape?
- A A straight line through the origin
- B A horizontal line
- C A hyperbola
- D A vertical line
Check answer
Answer: C. A hyperbola
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Frequently Asked Questions
What is the relationship between pressure and volume in Boyle's Law?
The relationship is inversely proportional, meaning as one variable increases, the other decreases at the same rate. This is valid only if the temperature and the amount of gas remain constant.
Why does Boyle's Law only apply to ideal gases?
Boyle's Law assumes that gas particles have no volume and no intermolecular forces, which are the characteristics of an ideal gas. Real gases deviate from this law at extremely high pressures or very low temperatures where particle interactions become significant.
How do you convert mmHg to atm?
To convert mmHg to atm, you divide the value in mmHg by 760. This is because 1 standard atmosphere is defined as exactly 760 mmHg or 760 torr.
Can Boyle's Law be used if the temperature changes?
No, Boyle's Law is specifically for isothermal processes where temperature is constant. If temperature changes, you must use the Combined Gas Law or the Ideal Gas Law.
What are some real-life applications of Boyle's Law?
Common applications include the mechanism of breathing in human lungs, the operation of a bicycle pump, and the way a syringe draws liquid. Scuba divers also use it to understand how pressure changes affect the air in their tanks and lungs at different depths.
Does the type of gas matter when using Boyle's Law?
For most introductory and advanced chemistry problems, the identity of the gas does not matter because all ideal gases behave the same way. Whether it is Oxygen, Nitrogen, or Helium, the P-V relationship remains consistent under standard conditions.
Want unlimited practice questions like these?
Generate AI-powered questions with step-by-step solutions on any topic.
Try Question Generator Free →Enjoyed this article?
Share it with others who might find it helpful.